Monday Oct. 15, 2007
The graded Experiment #2 reports were returned today. Revised
reports will be due in two weeks - on Mon. Oct. 29 Please return
your original report with your revised report.
Optional Assignment #4 (Controls of Climate) is due at the beginning of
class this coming Wednesday. Copies of the assignment are
available in PAS 588.
Optional Assignment #5 (Humidity) was handed out today and will be due
at the beginning of class next Monday, Oct. 22.
We will
first list the 4 humidity variables introduced in class last Friday.

This is a good place to put the answers to the in-class
optional assignment from last Friday.

Today we
will try to understand why it is possible to saturate air with
water vapor. Why is there an upper limit to the amount of water
vapor that air can contain? Why does this upper limit depend on
air temperature?
First we need to understand that the rate at which water evaporates
depends on the water's temperature.
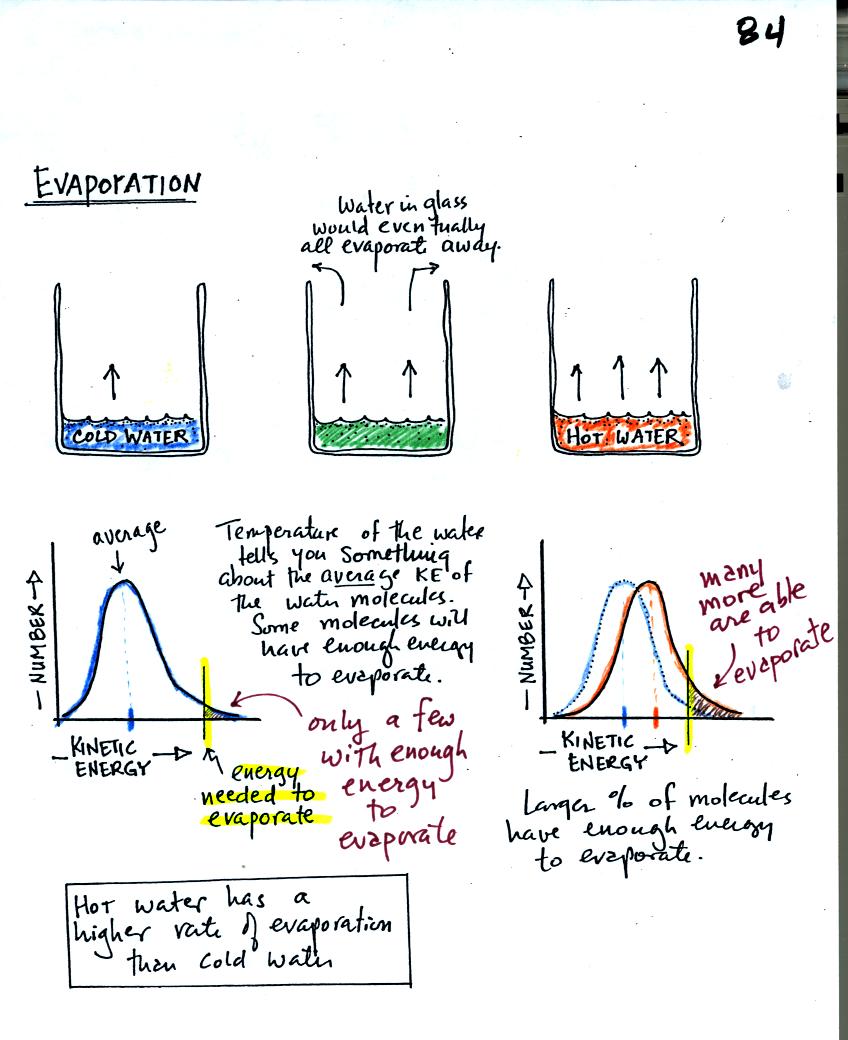
How water evaporates more rapidly than cold water. Picture a cup
of hot steaming coffee and a glass of iced tea.
To be able to evaporate, a water molecule in a glass must make its way
up to the
surface of the water and the water molecule must then have sufficient
kinetic energy
(to overcome any attractive forces trying to keep it in a liquid
state).
The distributions of the kinetic energies of the water molecules in the
glasses of cold and hot water (remember temperature is a measure of the
average kinetic energy) are shown in the two graphs above. In
cold water only a limited number of the water molecules
(those to the right of the highlighted line) have the necessary
energy - cold water has a low rate of
evaporation. In hot water, the whole distribution has moved to
the
right, the threshold energy needed to evaporate has remained the same,
and a larger fracton of the water
molecules will have enough energy. Hot water evaporates more
rapidly.
Now we will look at the top part of p. 85 in the photocopied
notes. We have put a cover on the glass of room temperature
water.
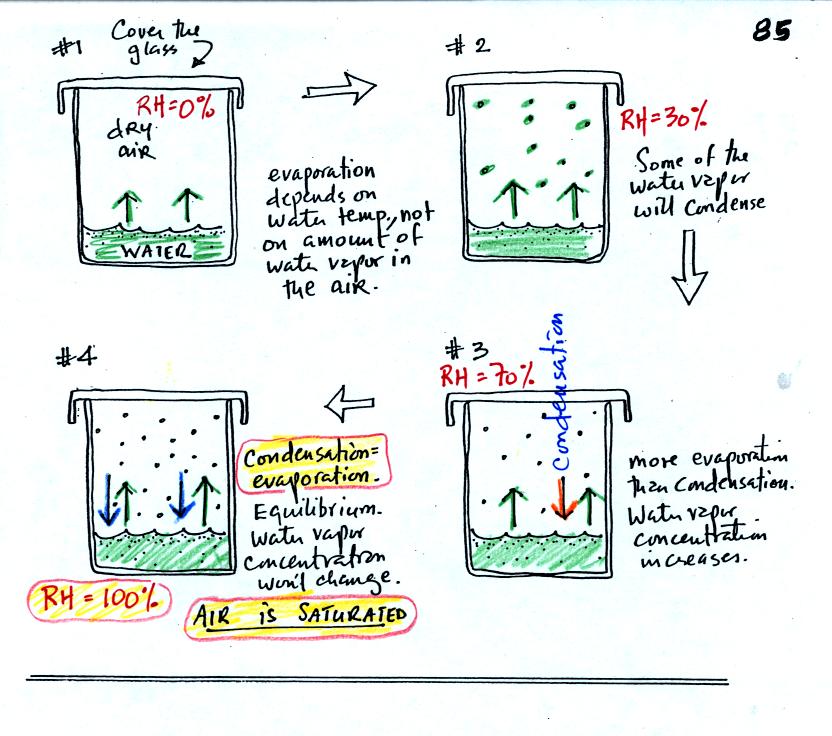
In #1 we see that the water is evaporating (2 arrows worth
of
evaporation). Water vapor will begin to build up in the air in
the glass. This is shown in #2. Some of the water vapor
molecules will condense (molecules that find themselves with lower than
average kinetic energy). In Fig. #3 the amount of water vapor has
built up to
a point where the amount of condensation is becoming significant and
one arrow worth of condensation has been added to the picture. In
#3
there is still more evaporation than condensation so the water vapor
concentration will increase a little bit more. Eventually in #4
the water vapor concentration has increased to a point that there are
two arrows of condensation. This balances the 2 arrows of
evaporation. The air is saturated, the air is filled to
capacity. With equal rates of evaporation and condensation, the
amount of water vapor in the air will now remain
constant. Note that the relative humidity is 100% at this point.
The air is filled to capacity.
What would happen if we took off the cover and added some
more water
vapor to the glass in Fig. #4? (this figure wasn't shown in class).
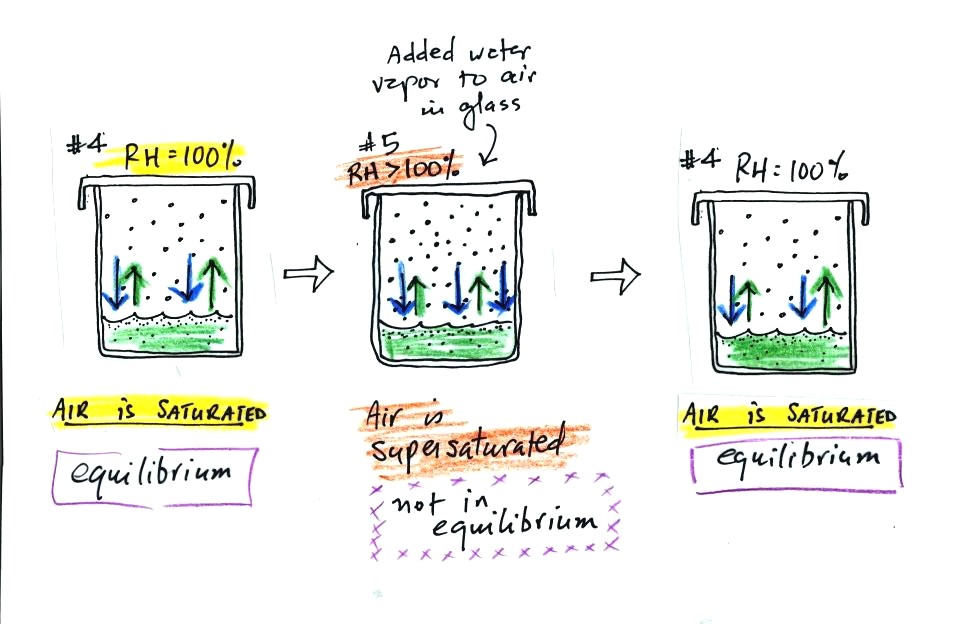
The air in Fig. #5 shows what would happen. The
air
would be supersaturated with water vapor and the RH would be greater
than 100%. This is possible but it is not an equilibrium
situation and wouldn't remain this way. The increased amount of
water vapor would increase the
rate of condensation. There would be more condensation than
evaporation. The water vapor concentration would begin to
decrease. Eventually the glass would return to the equilibrium
situation in Fig. #4.
If we look at the bottom of p. 85 we see that the air in all
three cases is
saturated (equal rates of evaporation and condensation in each
case). The relative humidity is 100% in all three cases.
But because the different rates of evaporation (in cold and warm water)
require different rates of condensation to be in balance, the of water
vapor in the air in the two cases however is different. The
warm air contains a lot more water vapor than the cold air.

At this point in class we took a short "break" and looked briefly
at Experiment #3.
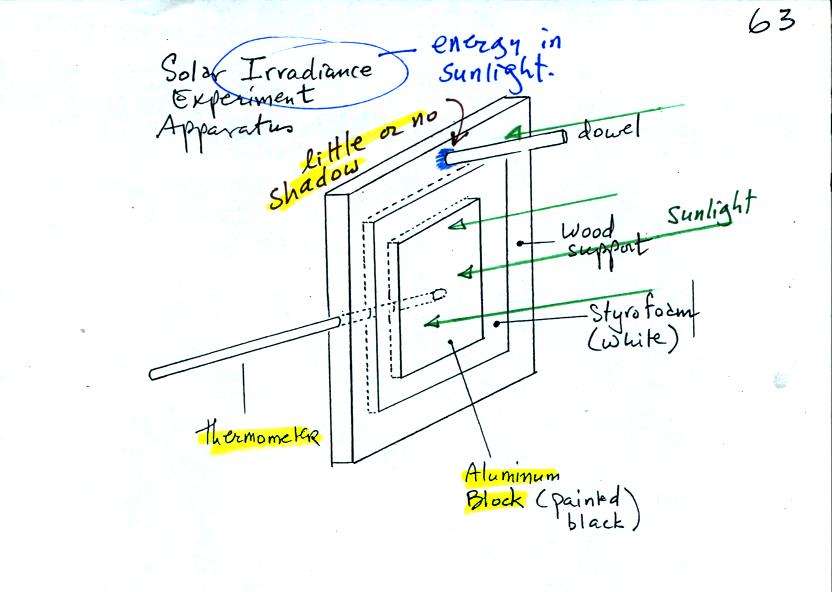
The object of Expt. #3 is to measure the energy in
sunlight. The
basic idea, shown in the figure below (see p. 63 in the photocopied
Class Notes) and in two short video tapes (that were hopefully much
clearer), is to point a flat piece of
aluminum (with known area, mass, and specific heat) straight at the sun
and measure how quickly it warms up.
Doesn't it seem reasonable to think that,
since it is the sunlight that is causing the aluminum to warm up
in the first place, you could use the temperature data to figure out
how much energy is in that sunlight? [the answer to that question
is Yes it does seem reasonable, very reasonable]
The problem is going from an idea that seems reasonable to an equation
that you can actually use. We didn't really work out all the
details in class but it's not that difficult. Click on this solar irradiance
link if you are interested in seeing the details.
Now back
to humidity
In the time remaining in class we were able to work a couple
of humidity example problems. By doing these problems (and we'll
do a couple more on Wednesday) you should become more
familiar with the humidity variables (mixing ratio, saturation mixing
ratio, relative humidity, and dew point temperature). You'll also
learn "how they behave" and what can cause each of these variables to
change value.
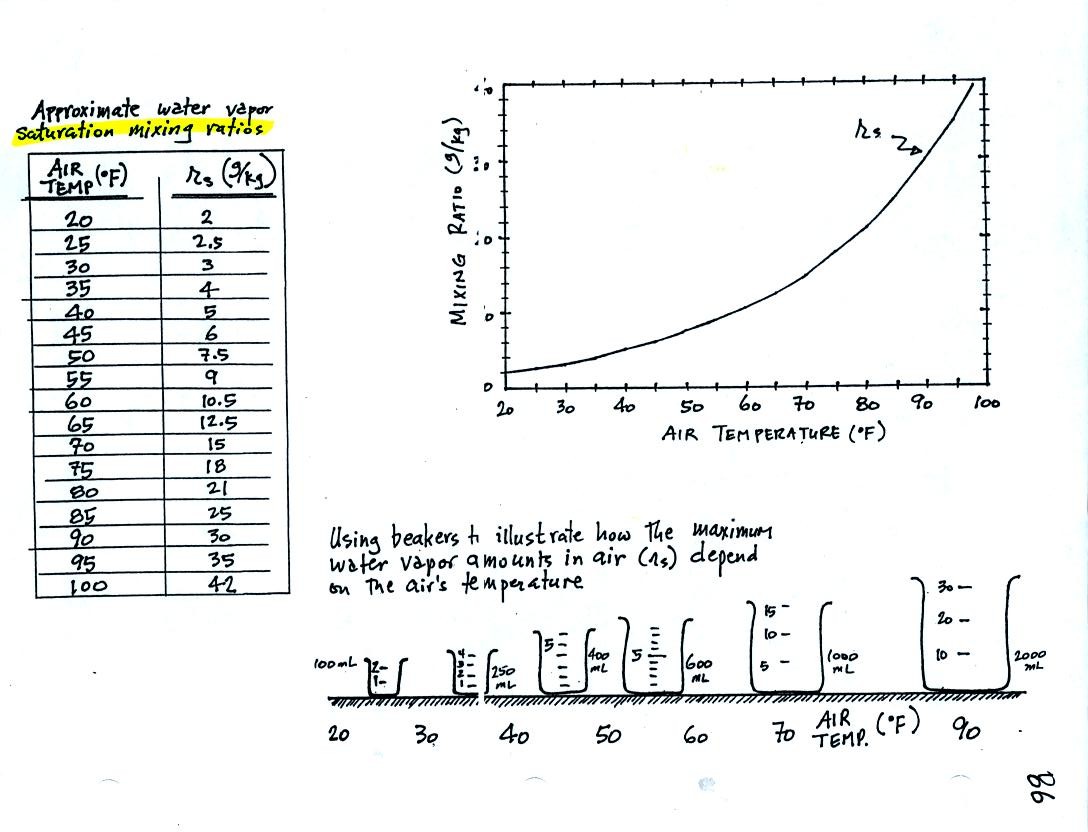
Keep this compilation of saturation mixing ratio values
(shown in a table and on a graph) handy, we will
use it a lot as we work through the humidity problem examples.
Remember that saturation mixing ratio is the maximum amount of water
vapor that can be found in air. It is a property of the air and
depends on the air's temperature.
The beakers (beakers were also brought to class)
are meant to show graphically the relative amounts of water
vapor that air at different temperatures can contain.
Now the
first of our example problems
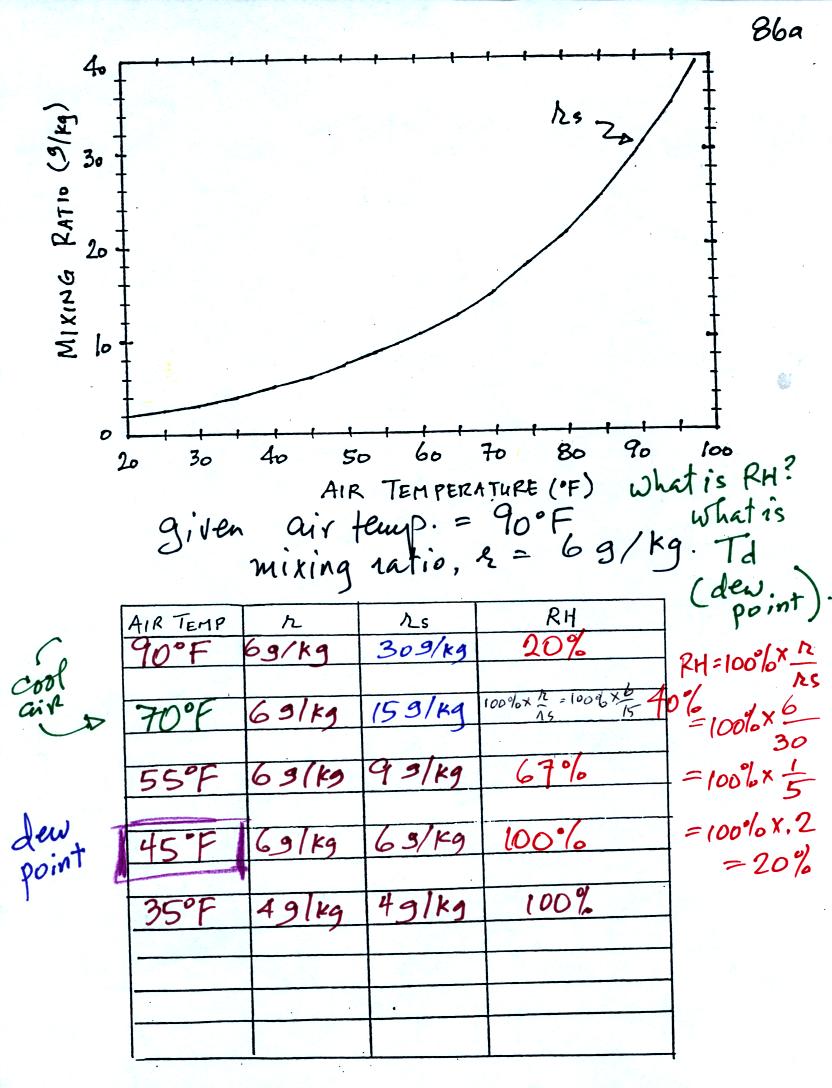
Here is the first sample
problem that we worked in
class.
You might have a hard time unscrambling this if you're seeing it for
the first
time. The series of steps that we followed are retraced
below:

We're given an air temperature of 90 F and a mixing ratio
(r) of 6
g/kg. We're supposed to find the relative humidity (RH) and
the dew point temperature.
We start by entering the data we were given in the
table. Once
you know the air's temperature you can look up the saturation mixing
ratio value; it is 30 g/kg for 90 F air. 90 F air could
potentially hold 30 grams of water vapor per kilogram of dry air (it
actually contains 6 grams per kilogram in this example).
Once you know mixing ratio and saturation mixing ratio you can
calculate the relative humidity. The RH is 20%.
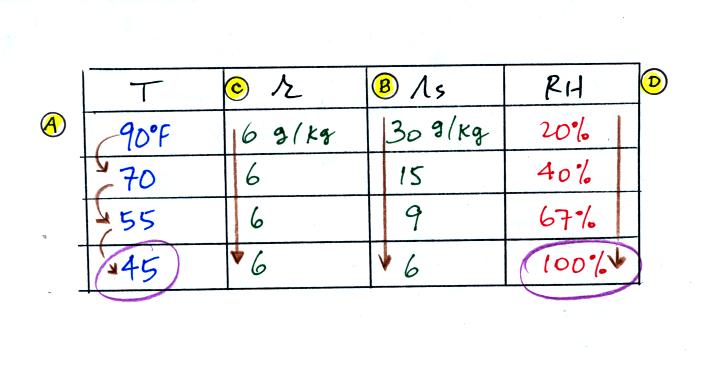
The numbers we just figured out are shown on the top line
above.
(A) We imagined cooling the air from 90F to 70F, then to 55F, and
finally to 45F.
(B) At each step we looked up the saturation mixing ratio and entered
it on the chart. Note that the saturation mixing ratio values
decrease as the air is cooling.
(C) The mixing ratio doesn't change as we cool the air. The only
thing that changes r is adding or removing water vapor and we aren't
doing either.
(D) Note how the relative humidity is increasing as we cool
the
air. The air still contains the same amount of water vapor it is
just that the air's capacity is decreasing.
Finally at 45 F the RH becomes 100%. The dew point temperature in
this problem is 45 F.
What would happen if we cooled the air further still, below the dew
point temperature?
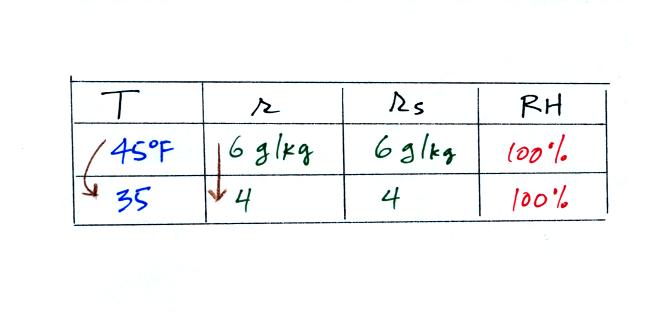
35 F air can't hold the 6 grams of water vapor
that 45 F air can. You can only "fit" 4 grams of water vapor into
the 35 F air. The remaining 2 grams would condense. If
this happened at ground level the ground would get wet with dew.
If it happens above the ground, the water vapor condenses onto small
particles in the air and forms fog or a cloud. Now because water
vapor is being taken out of the air (and being turned into water), the
mixing
ratio will decrease from 6 to 4. That is why the mixing ratio is
changing.
In many ways cooling moist air is liking squeezing a
moist sponge (the figure below
wasn't shown in class)
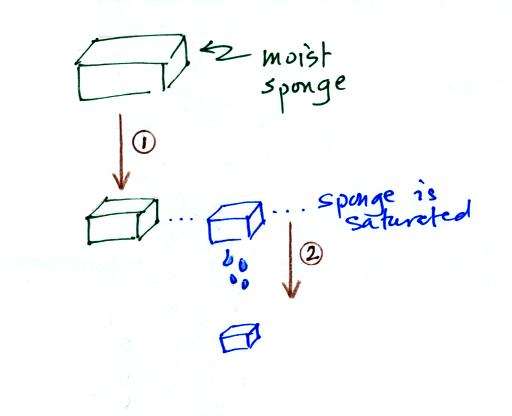
Squeezing the
sponge and reducing its volume is like cooling moist air and reducing
the saturation mixing ratio. At first when you sqeeze the sponge
nothing happens, no water drips out. Eventually you get to a
point where the sponge is saturated. This is like reaching the
dew point. If you squeeze the sponge any further (or cool air
below
the dew point) water will begin to drip out of the sponge (water vapor
will condense from the air).
Here's the
2nd problem we did
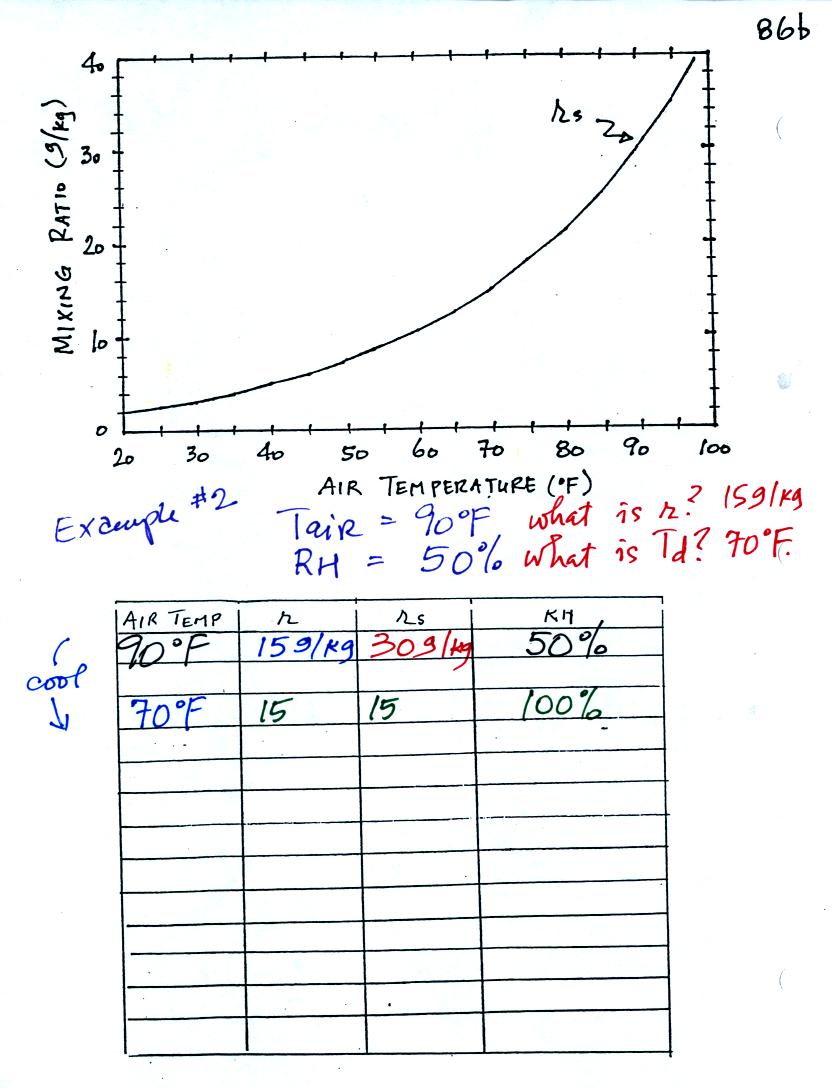
Here is what we did in class. Given an air
temperature
of 90
F and a relative humidity of 50% you are supposed to figure out the
mixing ratio (15 g/kg) and the dew point temperature (70 F). The
problem is worked out in detail below:

First you fill in the air temperature and the RH data that
you are
given.
(A) since you know the air's temperature you can look up the
saturation mixing ratio (30 g/kg).
(B) Then you can substitute into
the relative humidity formula and solve for the mixing ratio (15 g/kg).
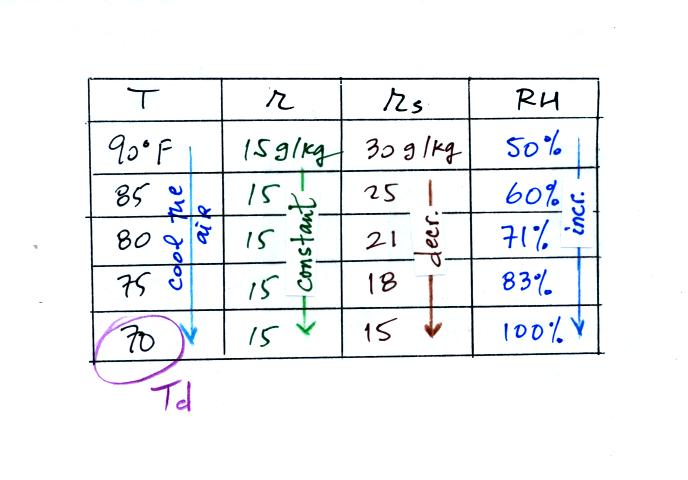
Finally you imagine cooling the air. Cooling causes
the
saturation mixing ratio to decrease, the mixing ratio stays constant,
and the relative humidity increases. In this example the RH
reached 100% when the air had cooled to 70 F. That is the dew
point temperature.















