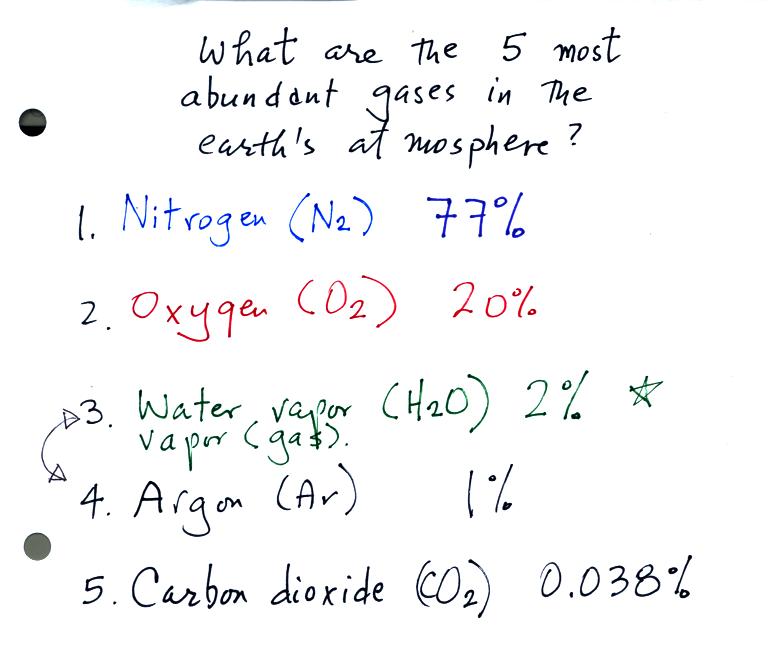Tuesday
Aug. 21
The
first day of class.
We first briefly discussed the Course
Information
handout. Note the various options you have for purchasing a copy
of the course textbook. You should try to purchase a copy of the
photocopied
Classnotes (in the bookstore) right away as we will probably be using
some of
them in class on Wednesday.
Next we looked at the Writing
Requirements
handout. You weren't given much time to think about which of four
experiments you would like to do before signup sheets were circulated
through class. Don't worry if you didn't sign up for an
experiment (or book report) there will be additional opportunities to
do so during the semester. Also there are often extra materials
when the experiment supplies are brought to class. You could
decide to check out materials at that time.
Your grade
in this class will depend on your quiz scores, how much
extra credit you earn, your writing grade, and (perhaps) your
score on the final exam. A sample grade report from the Spring
2007 T Th Nats 101 class was shown.
Example grade summary from the T Th class
last semester
(low Cs on all four quizzes, a D on the final exam, a C for the class)
Doe_J
quiz1 -42 (160 pts possible)
73.8%
quiz2 -42 (155 pts possible)
72.9%
quiz3 -50 (165 pts possible)
71.9%
quiz4 -44 (160 pts possible)
72.5%
2.8 EC points (3.3 pts possible)
writing scores: 33.5 (expt/book report) + 39.5
(1S1P pts)
writing grade: 91.3%
average (no quiz scores dropped): 76.5% + 2.8 =
79.3%
average (lowest quiz score dropped): 77.6% + 2.8 =
80.4
you DO need to take the final exam
-32.5 pts missed on the final exam = 64.3%
3Q&W>F overall average is 77.2
The MWF class
average (below) was a little higher than the T Th class last spring
(that is usually the case for some unknown reason, I hope that trend
changes this semester)
Doe_J
quiz1 -36 (160 pts possible)
77.5%
quiz2 -46 (155 pts possible)
70.3%
quiz3 -48 (165 pts possible)
73.1%
quiz4 -35 (160 pts possible)
78.1%
2.6 EC points (3.3 pts possible)
writing scores: 33.0 (expt/book report) + 45.0
(1S1P pts)
writing grade: 97.5%
average (no quiz scores dropped): 79.3% + 2.6 =
81.9%
average (lowest quiz score dropped): 81.6% + 2.6 =
84.2
you DO need to take the final exam
-24.8 pts missed on the final exam = 75.3%
3Q&W>F
overall average is 82.4
The
interesting thing here is that the quiz and final exam grades were all
Cs, but the overall grade for the class was a B. That is largely
due to the extra credit and the high writing grade.
Don't
worry about all the details on these grade summaries yet.
We'll
begin this new semester in Chapter 1 of the text. Before opening
the book and beginning the first reading
assignment, try to imagine
what you would put in the first chapter of a meteorology and
climatology textbook.

Student answers to the question above included whether there was
enough oxygen in the planet's air, what the temperature was, and what
the atmospheric pressure was. Many of
these are
covered in Chapter 1 of the text.
Today we
were concerned with the composition of the earth's atmosphere, in
particular the 5 most abundant gases in the earth's atmosphere.
Some of
the most abundant gas, in liquid form, was poured into a styrofoam cup.

The liquid was
nitrogen (you can fill in the blank above with the word
nitrogen). You can see liquid nitrogen, it is clear and looks
like
water. Once it has
evaporated and turned into a gas it is invisible.
Here's
a photograph of the second most abundant gas in the atmosphere in
liquid form.
Water vapor and argon are the 3rd and 4th most abundant
gases in the
atmosphere. The concentration of water vapor can vary from near
0% to as high as 3% or 4%. The variable concentration
of water vapor means it is sometimes more
abundant, sometimes less abundant than argon which has a concentration
of about 1%.
Here's the complete list that we came up with in class:
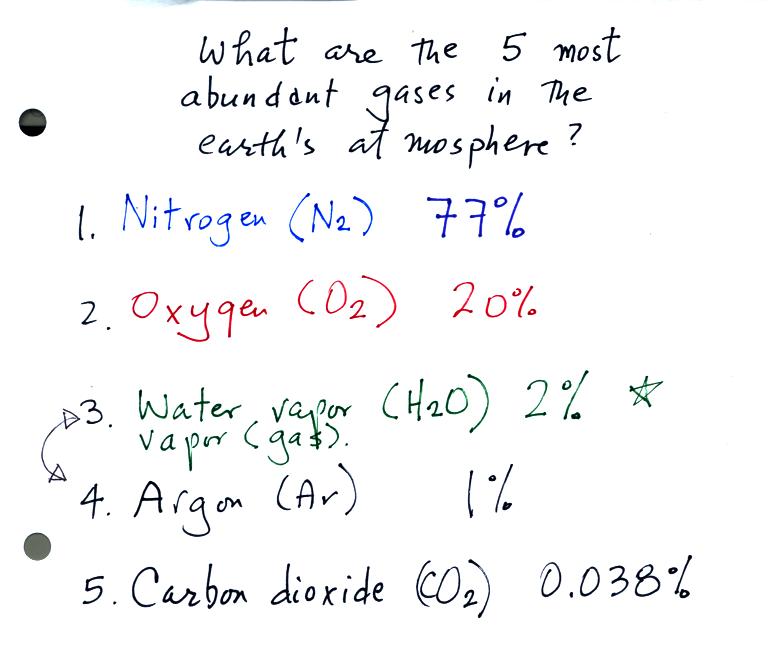
Water plays an important role in the formation of clouds,
storms,
and weather. Meteorologists are very interested in knowing and
keeping track of how
much water vapor is in the air at a particular place and time.
One of the variables they use is the dew point temperature; it has two
"jobs."

Its first job is to provide
a measure of the amount of water
vapor in the air. The dew point is just a number. When
the value
is low the air doesn't contain much moisture. The higher the dew
point value, the more water vapor there is in the air.
The chart below gives a rough equivalence between dew point
temperature and percentage concentration of water vapor in the air.
We are in the summer thunderstorm season in Tucson and dew
points
have been in the 60s. Many people use an evaporative cooler to cool their homes in
the summer. Evaporative coolers don't work very well when dew
points are as high as they are now.
Click here
to see current dew point temperatures across the U.S. Note that a
large part of the SE US currently has dew points in the 70s.
The second job of the dew point temperature is illustrated
below. When you cool moist air to its dew point, the
relative humidity becomes 100% and a cloud forms.

The
following information was covered briefly at the end of class.
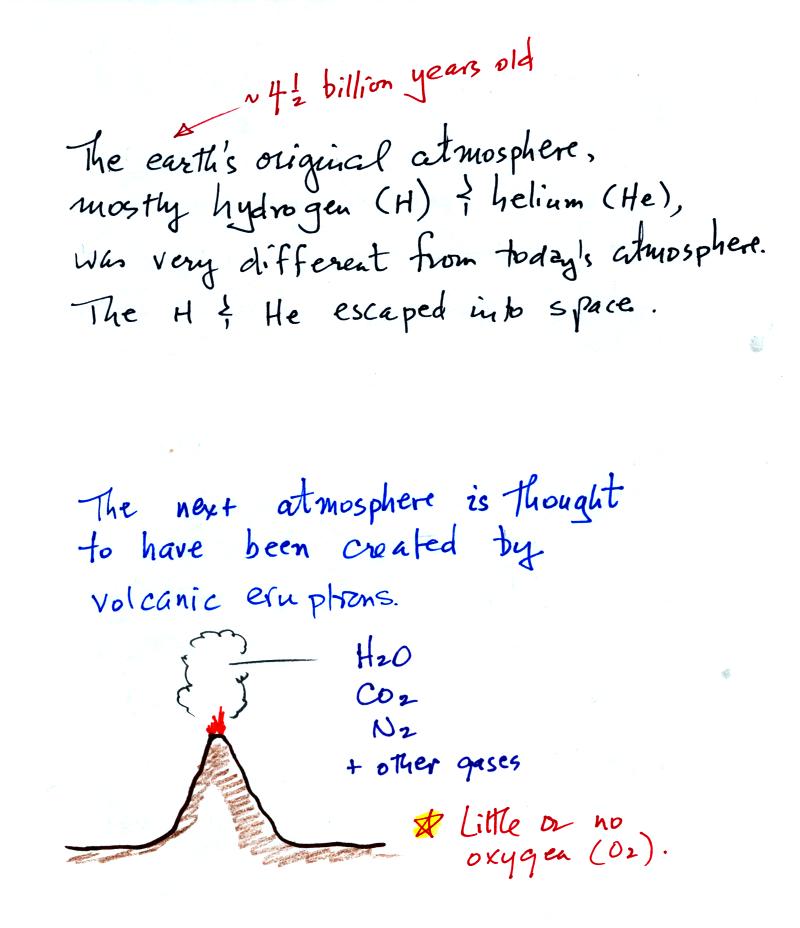
The earth's first atmosphere was composed mainly of hydrogen
and
helium.
These light-weight gases escaped into space and were lost. The
next atmosphere was built up of gases emitted during volcanic
eruptions, mostly water vapor, carbon dioxide, and nitrogen. As
the earth began to cool the water vapor condensed and began to create
oceans. Carbon dioxide dissolved in the oceans and was slowly
turned into rock. Much of the nitrogen remained in the atmosphere.
Note the volcanoes didn't add oxygen to the atmosphere.

The oxygen is thought to have first come from
photodissociation of
water vapor and carbon dioxide by ultraviolet light.. Photodissociation
is the splitting apart of the H20 and CO2
molecules by the ultraviolet light. The O and OH pieces react
to form O2 and H.
Unfiltered ultraviolet light from the sun would be deadly to plant and
animal life on the earth's surface. Once O2 began to
accumulate in the air, O3, began to form and buildup in the
atmosphere. Ozone began to absorb most of the harmful
ultraviolet
light before it reached the surface and life forms were able to move
from the oceans (which would absorb UV light in the
absence of ozone) onto land.
Eventually plants and photosynthesis
would become the main source of atmospheric oxygen.
Here are
some other gases found in the earth's atmosphere that we
will cover (this list was not
covered in class). Most are found in very low
concentrations but that
doesn't
mean they are not important.
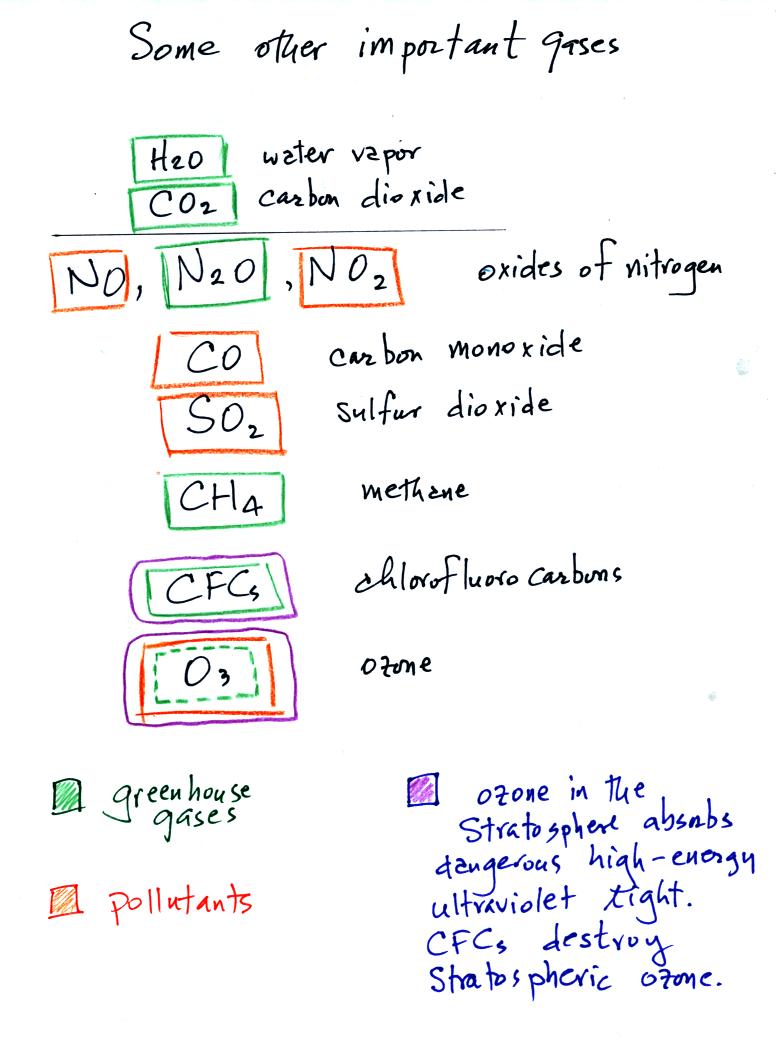
Water vapor, carbon dioxide, methane, nitrous oxide (N2O
=
laughing gas),
chlorofluorocarbons, and ozone are greenhouse gases. We'll
discuss the greenhouse effect a little bit more later in class today
and will
learn more about it actually works when we get to Chapter
2.
Carbon monoxide, nitric oxide, nitrogen dioxide, ozone, and sulfur
dioxide are some of the major air pollutants. We'll cover 2 or 3
of these in class on Tuesday next week. You may have
heard or read about an incident earlier this week where carbon monoxide
from a malfunctioning hot water heater sickened 23 Virginia Tech
students in an apartment complex. Carbon monoxide
concentrations indoors can easily and rapidly reach fatal levels.
Carbon monoxide
levels in the atmosphere are much lower but can still represent a
health hazard.
Ozone in the stratosphere (a layer of the atmosphere between 10 and 50
km) is beneficial because it absorbs dangerous high energy ultraviolet
(UV) light coming from the sun. Without the protection of the
ozone layer life as we know it would not exist on the surface of the
earth. Chlorofluorocarbons are of concern in the atmosphere
because they destroy stratospheric ozone. In the
troposphere (the bottom 10 kilometers of the atmosphere) ozone is a
pollutant and is one of the main ingredients in photochemical smog.