Friday Oct. 7, 2011
click here to
download today's notes in a more printer friendly format
I got distracted and started the music a little late.
There was time for only on Domingo
DeGrazia song ("Tres
Ninas")
The Upper Level Charts Optional Assignment has been graded and was
returned in class. Everyone that did the assignment received 0.5
pts of extra credit. If you missed 3 pts or less you also earned
a Green Card.
You'll find a link on the class home page that shows you how to estimate your grade
in this class. I will try to hand out grade summaries after Quiz
#2 and once we have all the 1S1P Assignment #1 papers graded.
In class on Wednesday we learned
about the laws that govern the amount and kind of EM radiation emitted
by an object. We looked at the light produced by an ordinary
tungsten bulb as an example.
Now we will compare the light emitted by the sun and the earth.
The curve on the left is for the sun. We first used Wien's
law and a temperature of 6000 K to calculate λmax
and got
0.5 micrometers. This is green light; the sun emits more green
light than any other kind of
light. The sun doesn't appear green because it is also emitting
lesser amounts of violet, blue, yellow, orange, and red - together this
mix of
colors appears white. 44% of the radiation emitted by the sun is
visible light, 49% is IR light (37% near IR + 12% far IR), and 7%
is ultraviolet light. More than half of the light emitted by the
sun is invisible.
100% of the light emitted by the earth (temperature = 300 K) is
invisible IR light. The
wavelength of peak emission for the earth is 10 micrometers.
Because the sun (surface of the
sun) is 20 times hotter than the earth the sun's
surface emits energy at a much higher rate than the earth. Note
the
vertical
scale
on the earth curve is different than on the sun
graph. If both the earth and sun were plotted with the same
vertical scale, the earth curve would be too small to be seen.
On Wednesday
we also learned that ordinary tungsten bulbs (incandescent
bulbs) produce a lot of
wasted energy. This is because they emit a lot of invisible
infrared light that doesn't light up a room (it will warm up a room but
there are better ways of doing that). The light that they do
produce is a warm white color (tungsten bulbs emit lots of orange, red,
and yellow light and not much blue, green or violet).
Energy
efficient
compact
fluorescent
lamps
(CFLs)
are
being
touted
as
an
ecological
alternative
to
tungsten bulbs because
they use substantially less electricity, don't emit a lot of
wasted infrared light, and also last
longer. CFLs come with
different color temperature ratings.
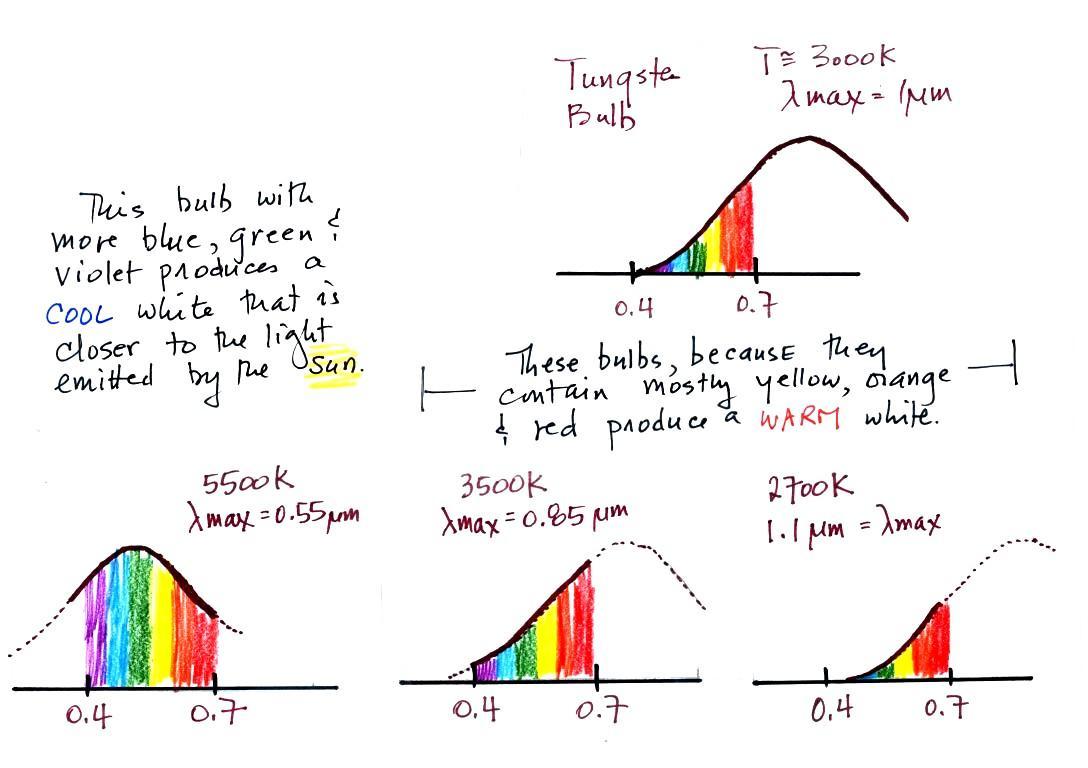
The bulb with the hottest
temperature rating (5500 K ) in the figure
above is meant to mimic or simulate sunlight. The temperature of
the sun is 6000 K and lambda max is 0.5 micrometers. The spectrum
of the 5500 K bulb is similar.
The tungsten bulb (3000 K) and the CFLs with temperature ratings
of
3500 K and 2700 K produce a warmer white.
Three CFLs with the temperature ratings above were set up in class
so
that you could see the difference between warm and cool white
light. Personally I find the 2700 K bulb "too warm," it makes a
room
seem gloomy and depressing (a student once class the
light resembles Tucson at night). The 5500 K bulb is "too cool"
and
creates
a stark sterile atmosphere like you might see in a
hospital corridor. I prefer the 3500 K bulb in the
middle.
The figure below is from an article
on compact fluorescent lamps in Wikipedia for those of you that weren't
in class and didn't see the bulb display.. You can
see a clear difference between the cool white bulb on the left
in the figure below and the warm white light produced by a tungsten
bulb (2nd from the left) and 2 CFCs with low temperature ratings (the 2
bulbs at right).
There is one downside to these energy efficient CFLs. The
bulbs
shouldn't just be discarded in your ordinary household trash because
they contain mercury. They should be disposed of properly (at a
hazardous materials collection site or perhaps at the store where they
were purchased).
It probably won't be long before LED bulbs begin to
replace tungsten and CFL bulbs. At the present time the LED bulbs
are pretty expensive.
We now
have most of the tools we will need to begin to study energy balance on
the earth. It will be a balance between incoming sunlight
energy and outgoing energy emitted by the earth. This will
ultimately lead us to an explanation of the atmospheric greenhouse
effect.
We will first look at the simplest kind of situation, the earth without
an atmosphere (or at least
an atmosphere without greenhouse gases). The next figure is on p.
68 in the
photocopied Classnotes. Radiative equilibrium is really just
balance between incoming and outgoing radiant energy.
You might first wonder how it is possible for the earth (with a
temperature
of around 300 K) to be in energy
balance with the sun (6000 K). At the top right of the figure you
can see that because the earth is located about 90
million miles
from the sun and it only absorbs a very small fraction of the
total energy emitted by the sun.
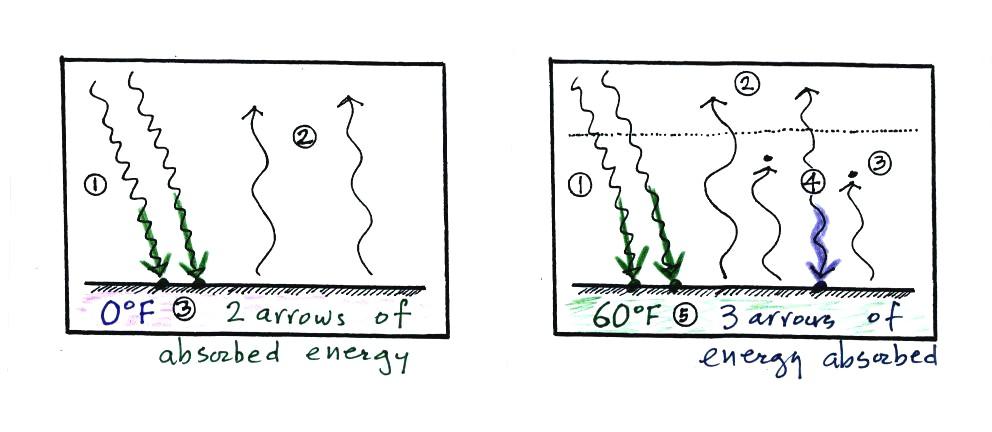
To understand how energy balance occurs we start, in Step #1, by
imagining that the earth starts out very cold (0 K) and is
not emitting
any EM radiation at all. It is absorbing sunlight however (4
of
the 6
arrows of incoming sunlight in the first picture are absorbed, 2 of the
6 are being reflected) so it
will
begin to warm This is like opening a bank account, the balance
will be zero at first. But then you start making deposits and the
balance
starts to grow.
Once the earth starts to warm it will also begin to emit EM
radiation, though not as much as it is getting from the sun (the
slightly warmer earth in the middle picture is now colored blue).
Only the four arrows of incoming sunlight that are absorbed are shown
in the
middle figure. The two arrows of reflected sunlight have been
left off because they don't really play a role in energy balance.
Reflected sunlight is like a check
that bounces. It really doesn't affect your bank account
balance. The earth is emitting 3 arrows of IR light
(in red). Because the earth is still gaining more
energy than it is losing the
earth will warm some more. Once you
find money in your bank account you start to spend it. But as
long as deposits are greater than the withdrawals the balance will grow.
Eventually it will warm enough that the earth (now shaded brown
& blue)
will
emit the same amount
of energy as it absorbs from
the sun. This is radiative equilibrium, energy balance (4 arrows
of absorbed energy are balanced by 4 arrows of emitted energy).
The
temperature at
which this occurs is about 0 F.
That is called the temperature of radiative equilibrium.
Note that it is the amounts of energy not the kinds of energy that
are important. Emitted radiation may have a different wavelength
than
the absorbed energy. That doesn't matter. As long as the
amounts are
energy the earth will be in energy balance.
Before we
start to look at radiant energy balance on the earth with an atmosphere
we
need to learn about filters. The atmosphere will filter sunlight
as it
passes through the atmosphere toward the ground. The atmosphere
will
also filter IR radiation emitted by the earth as it trys to travel into
space.
We will first look at the effects simple blue, green, and red glass
filters have on visible light. This is just to be able
to interpret a filter absorption curve or graph.
If you try to
shine white light (a
mixture of all the colors) through a
blue filter, only the blue light passes through. The filter
absorption curve shows 100% absorption at all but a narrow range of
wavelengths that correspond to blue light. The location of the
slot or gap in the absorption curve shifts a little bit with the green
and red filters.
The following figure is a simplified, easier to
remember,
representation of the
filtering effect of
the atmosphere on UV, VIS, and IR light (found on p. 69 in the
photocopied notes). The figure was redrawn after class.
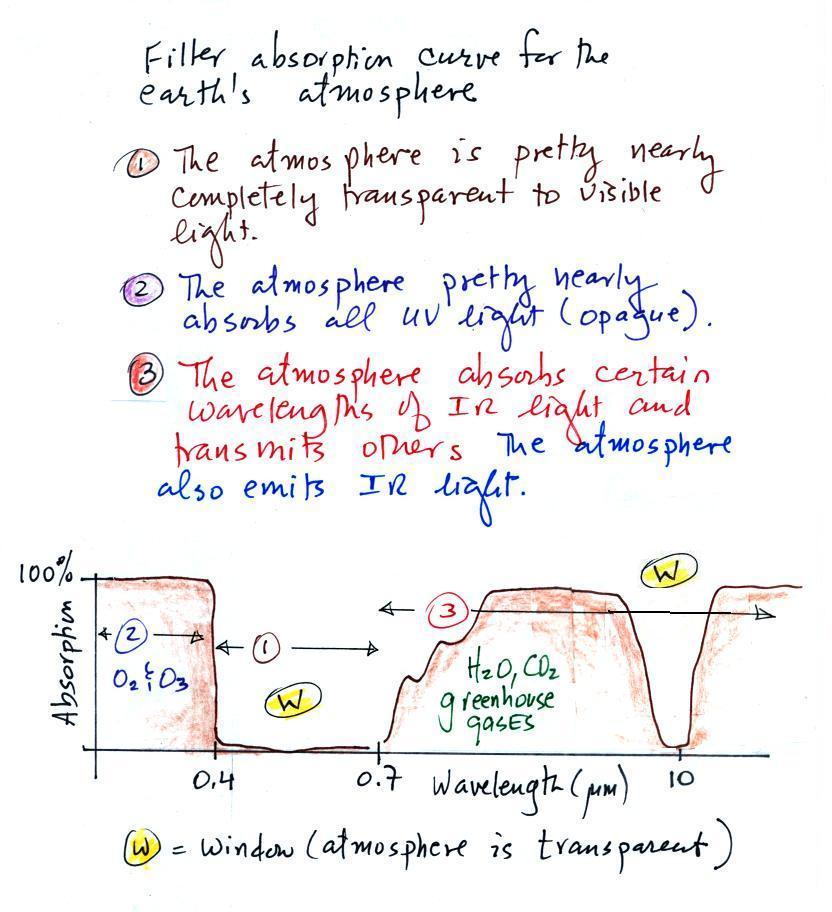
You can use your own eyes to tell
you what the filtering
effect of the
atmosphere is on visible light. Air is clear, it is
transparent. The atmosphere transmits visible light.
In our simplified representation oxygen and ozone make the
atmosphere pretty nearly completely opaque to UV light (opaque is the
opposite of transparent and means
that light is blocked or absorbed; light can't pass through an opaque
material). We
assume that the
atmosphere absorbs all incoming UV light, none of it makes it to the
ground. This is of course not entirely realistic.
Greenhouse gases make the
atmosphere a
selective absorber of IR light - the air absorbs certain IR wavelengths
and
transmits others. It is the atmosphere's ability to absorb (and
also emit) certain wavelengths of infrared light that produces the
greenhouse effect and warms the surface of the earth.
Note "The atmospheric window"
centered at 10 micrometers. Light emitted by the earth
at this
wavelength (and remember 10 um is the wavelength of peak emission for
the earth) will pass through the atmosphere. Another transparent
region, another window, is found in the visible part of the spectrum.
You'll find a more realistic picture of the atmospheric absorption
curve on p. 70 in the photocopied Classnotes, but the simplified
version above will work fine for us.
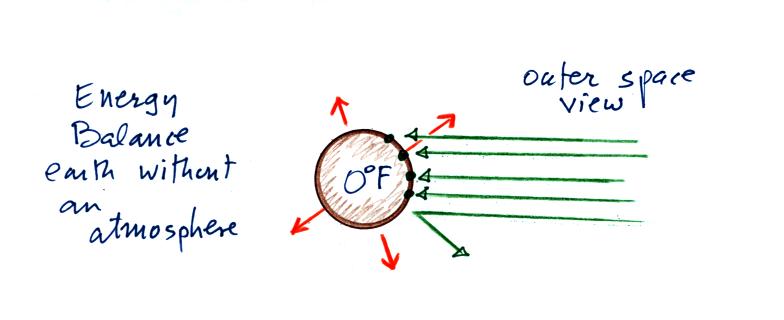
Here's the
outer space view of radiative equilibrium on the earth without an
atmosphere. The important thing to note is that the earth is
absorbing and emitting the same amount of energy (4 arrows absorbed
balanced by 4 arrows emitted).
We will be moving from an outer
space vantage point of
radiative equilibrium (figure above) to the earth's
surface (the next two figures below).
Don't let the fact that there are
4 arrows are
being absorbed and
emitted in the top figure and
2 arrows absorbed and emitted in the bottom figure
bother you. The important
thing is that there are equal
amounts being absorbed and emitted in both cases.
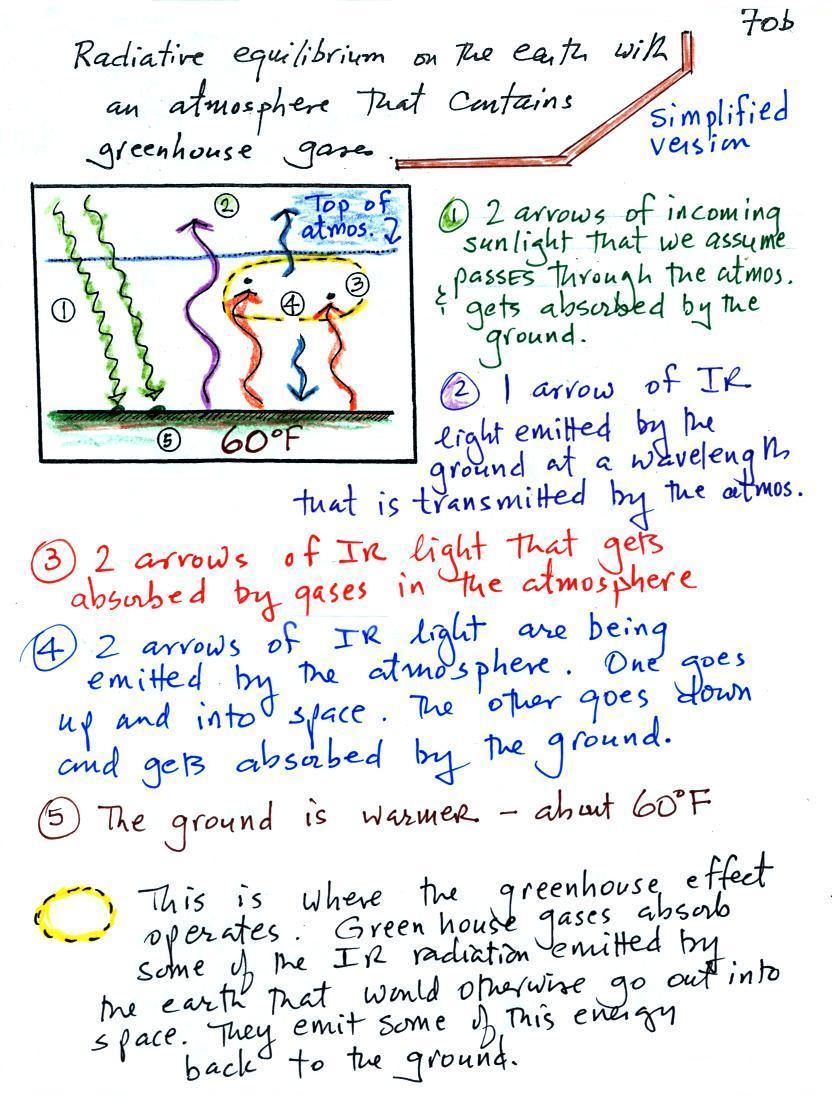
Here's the same picture with some
information added (p. 70a in the photocopied ClassNotes). This
represents energy balance on the earth without an atmosphere.
The next
step is to add the atmosphere.
We will study a simplified
version
of radiative equilibrium just so you
can identify and understand the various parts of the picture.
Keep an eye out for the greenhouse effect. Here's a cleaned
up version of what
we ended up with in class (I added a little information at the bottom
of the picture.
It would be hard to sort through and try to understand all of this
if you weren't in
class
(probably even more difficult if you were in class). So below we
will go through it again step by step (which you are free to skip over
if you wish). This is a more
detailed version than was done in class. Caution:
some of the colors below are different
from those used in class.
1. In this
picture we see the two
rays of incoming sunlight that
pass through the atmosphere, reach the ground, and are absorbed.
100% of the incoming sunlight is transmitted by the atmosphere.
This wouldn't be too bad of an assumption if sunlight were just visible
light. But it is not, sunlight is about half IR light and some of
that
is going to be absorbed. But we don't worry about that at this
point.
The ground is emitting
a total of 3 arrows of IR radiation.
2. One
of
these
(the
pink
or
purple
arrow
above)
is
emitted
by
the
ground at a wavelength
that is
NOT ABSORBED by greenhouse gases in the atmosphere (probably around 10
micrometers). This
radiation passes through the atmosphere and goes out into space.
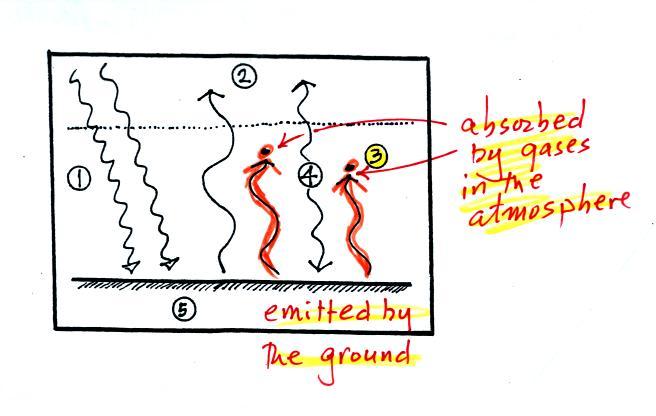
3. The other 2
units of IR radiation emitted by
the
ground are
absorbed by
greenhouse gases is the atmosphere.
4. The
atmosphere is absorbing
2 units of radiation.
In order to be in radiative equilibrium, the atmosphere must also emit
2
units of radiation. That's shown above. 1
unit of IR radiation is sent upward into space, 1 unit is sent downward
to the ground where it is absorbed. This is probably the part of
the picture that most students have trouble visualizing (it isn't so
much that
they have trouble understanding that the atmosphere emits radiation but
that 1 arrow is emitted upward and another is emitted downward toward
the ground.
Before we go any further we will
check
to be sure that
every part
of this picture is in energy balance.
The ground is absorbing
3 units of energy (2 green
arrows of sunlight and one bluish arrow coming from the atmosphere) and
emitting
3
units of energy (one pink and two red arrows). So the ground is
in energy balance.
The atmosphere is
absorbing 2 units of energy (the 2
red arrows coming from the ground) and
emitting 2
units of
energy (the 2 blue arrows). One goes upward into space. The
downward arrow goes all the way
to the ground where it gets absorbed (it leaves the atmosphere and gets
absorbed by the ground). The atmosphere is in energy balance.
And we should check to be sure equal amounts of energy
are arriving at and leaving the earth. 2 units of energy arrive
at the top of the atmosphere (green) from the sun after traveling
through space, 2 units
of
energy (pink and orange) leave the earth and head back out into
space. Energy balance here too.
The greenhouse effect involves the absorption and
emission
of IR radiation by the atmosphere. Here's how you might put it
into words (something
I
didn't do in class on Friday):

Doesn't it make sense that if the
ground is getting back some of
the energy it would otherwise lose, the ground will end up being
warmer. That's what the greenhouse effect does, it warms the
earth's surface. The global annual average surface temperature is
about 60
F on the earth with a greenhouse effect. It would be about 0 F
without the greenhouse effect.
Here are a couple other ways of understanding why the greenhouse
effect warms the earth.
The picture at left is
the earth without an atmosphere (without a greenhouse effect). At
right the earth has an
atmosphere, one that contains greenhouse gases.
At left the ground
is getting 2 units of energy (from the sun). At right it is
getting three, two from the sun and one from the atmosphere (thanks to
the greenhouse effect).
Doesn't it seem
reasonable
that ground that absorbs 3 units of energy will be warmer than ground
that is only absorbing 2?
Here's another explanation of why the ground is warmer with a
greenhouse effect than without.
At left the ground is emitting 2 units of energy, at
right the ground is emitting 3 units. Remember that the amount of
energy emitted by something depends on temperature. The ground
in the right picture must be warmer to be able to emit 3 arrows of
energy rather than 2
arrows. It is able to emit 3 arrows of energy even though it only
gets 2 arrows of sunlight because it is able to get a 3rd arrow of
energy from the atmosphere.