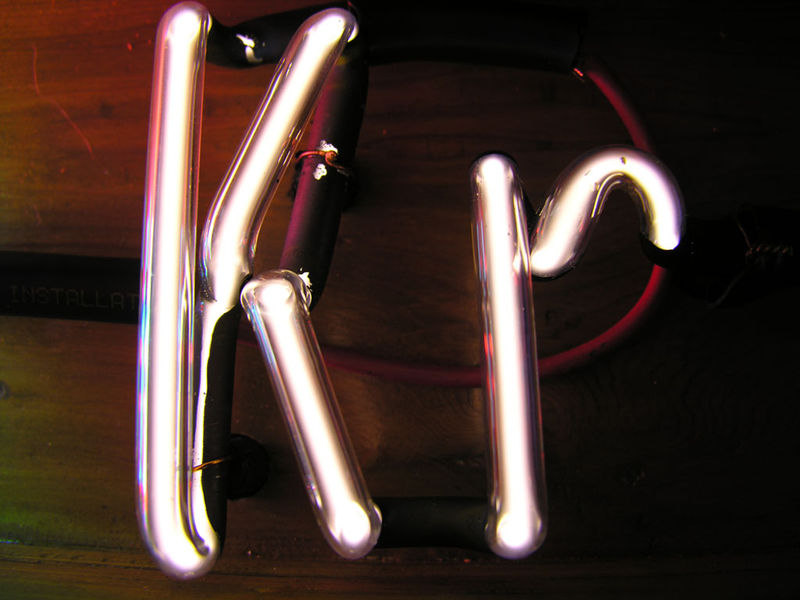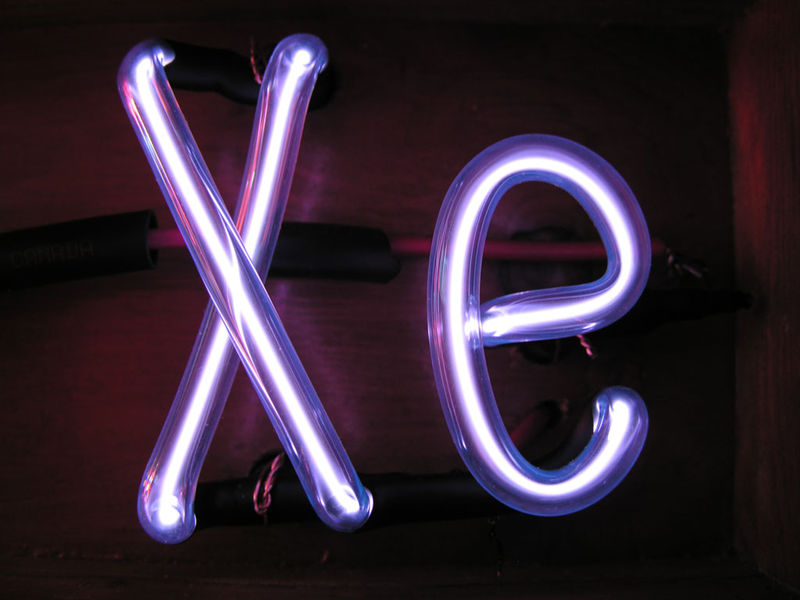Tuesday Aug. 27, 2013
click here to download a more
printer friendly version of today's notes
Music from Laura Marling, Mumford & Sons and Dharohar
Project ( Devil's
Spoke and Meheni Rachi
) to get another semester of ATMO 170 off to a proper start.
I discovered them by accident this summer after reading about
Laura Marling in RollingStone magazine and doing a search on
YouTube. I also really liked Laura
Marling's solo version of Blackberry Stone.
I'll try to play a lot of different kinds of music before
class. Hopefully you'll like at least some of it.
Suggestions from students are always welcome.
Today was the first day of class. We first
briefly discussed the Course
Information handout. Please read through that information
carefully on your own and let me know if you have any
questions.
A textbook is not required for this class. If you want to
get a more complete picture of the subject than we will have time
to do in class, you might want to purchase one of the textbooks
that are being used in the other ATMO 170A1 sections. Or if
you'd like to borrow one of the copies
of introductory level textbooks that I have in my office,
just let me know. Otherwise you should be able to do
perfectly well in the class by reading the online notes.
Read through the online notes even if you are in class.
A set of photocopied ClassNotes (available in the ASUA
Bookstore in the Student Union) is required. You should try
to purchase a copy as soon as you can because we will probably be
using the first page in class on Thursday. If you know
someone with a set of ClassNotes from the Fall 2012 or Spring 2013
class they should work fine this semester also.
Writing is an important part of
this class and is described in more detail on the Writing Requirements handout.
Please have a careful look
at that also and
let me know if you have any questions.
The first half of your writing grade is an experiment
report. You only need to do one of the experiments, so think
about which of the experiments (listed on the handout) you might
like to do. I'll bring a signup sheet to class on
Thursday. I'm also planning on bringing about 40 sets of
materials for the first experiment on Thursday.
The so-called One Side of One Page (1S1P) reports make up the
second part of your writing grade. Topics will appear
periodically during the semester on the class webpage. As
you write reports you will earn points (the exact number of points
will depend on the topic and the quality of your report).
Your goal should be to earn 45 1S1P pts, the maximum number
allowed, by the end of the semester.
You'll be allowed to revise and raise your grade on the first
draft of your experiment report. So you should be able to
earn a pretty high score on that. And, unless you
procrastinate, you can just keep on writing 1S1P reports until
you've earned 45 points. There's no reason not to earn a
high writing grade.
Your final grade in this class will depend on your quiz scores,
how much extra credit you earn (from optional take home and in
class assignments), your writing grade, and (perhaps) your score
on the final exam. A sample grade report from the Fall 2012
class is shown below (the numbers are class averages).
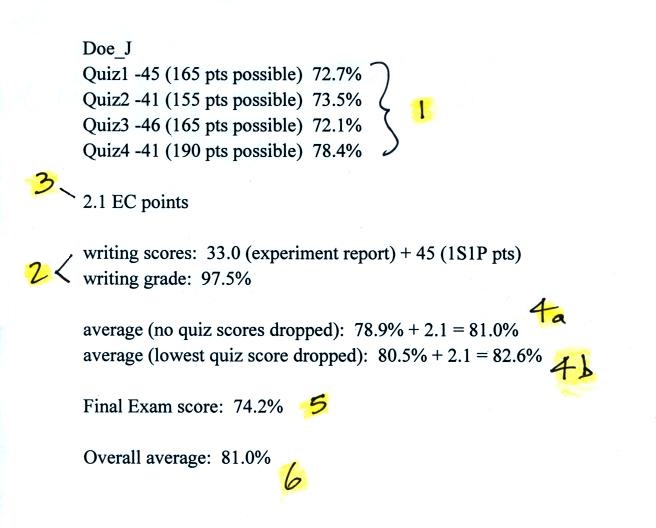
The 4 quiz grades are shown at Point 1.
At Point 2 you can see that this average student scored 33 out
of 40 on the experiment report and earned 45 1S1P pts (the
maximum number possible). This resulted in a writing
percentage grade of 97.5%.
The average student earned 2.1 extra credit points (you will
have the opportunity to earn at least 3 extra credit
points). This is shown at Point 3.
Point 4a shows the average of the 4 quizzes and the writing
grade with the extra credit points added on. In this case
the average, 81.0%, was less than the 90.0% or above needed to
get out of the final exam. Because the average at Point 4a
is less than 90.0% a second average with the lowest quiz score
dropped is computed, it is shown at Point 4b.
If you do well on the final exam it will count 40% of your
overall grade (trying to maximize the benefit it can
have). If you don't do so well on the final it only counts
20% (minimizing the damage it can cause). In this example
the final exam score (74.2%) was lower than the 82.6% value at
Point 4b, so the final only counted 20%. The overall score
ended up 81.0%. So even though this student had C grades
on all 4 quizzes and the Final Exam, the student ended up with a
B in the class.
We did cover a little course material in
class today just so you can get an idea of how that will work.
If we were using a book we'd
start in Chapter 1 and here's some of what we would be looking
at first in this course.
We had enough time today to look at the first question, the
composition of the atmosphere.
Before we do that however, here are a few questions to get you
thinking about the air around you. This is an example of extra information that I stick in the online
notes even though we didn't cover it in class.
That's something I warned you about in class.
Can
you see air?
Air
is
mostly clear, transparent, and invisible (that would be true
of the air in the classroom). Sometimes the air looks
foggy, hazy, or smoggy. In these cases you are "seeing"
small water droplets or ice crystals (fog) or small particles
of dust or smoke (haze and smog). The particles
themselves may be too small to be seen with the naked eye but
are visible because they scatter (redirect) light. I
didn't really mention or explain what that is but it's a
pretty important concept and we will learn more about it in a
week or so.
And actually air isn't really invisible. If you shine a
bright light through enough air, such as when sunlight shines
through the atmosphere, the air (the sky) appears blue.
This is a little more complicated form of scattering of
sunlight by air molecules. We'll come back to this later
as well.
Can you
smell air?
I don't think you can smell or
taste air (air containing nitrogen, oxygen, water vapor, argon
and carbon dioxide). But there are also lots of other odors
you can sometimes smell (freshly cut grass, hamburgers on a
grill, etc). I don't consider these normal constituents of
the atmosphere.
You can probably also smell certain pollutants. I suspect
our sense of smell is sensitive enough for us to detect certain
air pollutants even when their concentration is very small
(probably a good thing because many of them are
poisonous).
Natural gas (methane) used in
hot water heaters, some stoves, and furnaces is odorless.
A chemical (mercaptan) is added to natural gas so that you can
smell it and know when there is a leak before it builds up to a
concentration that could cause an explosion.
Can you feel air
It is harder to answer this question.
We're always in contact with air. Maybe we've grown so
accustomed to it we aren't aware of how it feels. We can
certainly feel whether the air is hot or cold, but that have
more to do with energy exchange between us and our
surroundings. And we can feel wind.
In a week or two we will see that, here in the classroom, air
pressure is pressing on every square inch of our bodies with 12
or 13 pounds of force. If that were to change suddenly I'm
pretty sure we'd feel it and it would probably really hurt.
Now back to material we did cover in class.
What are the 5 most abundant gases in
air?
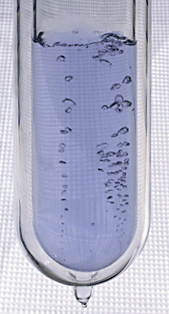
Let's start with the most abundant gas in the atmosphere.
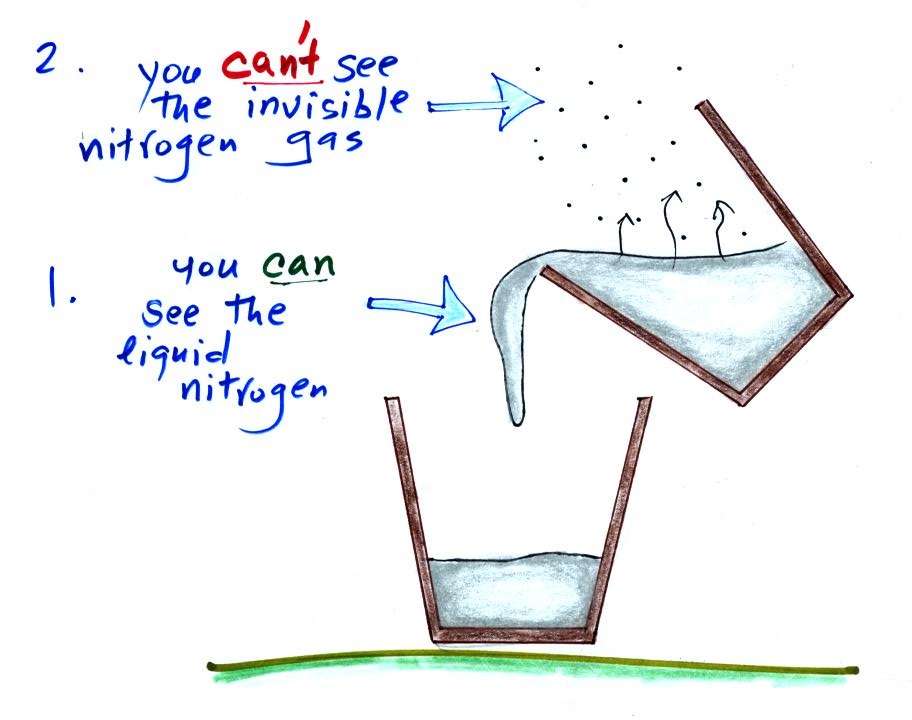
I poured some of this same material (in liquid form) into a
Styrofoam cup. Here's a photo I took back in my office.
You can see the liquid, it's clear, it looks like water.
At least one student (probably many more) knew that this
was liquid nitrogen.
The most abundant gas in the atmosphere is nitrogen. We'll
use liquid nitrogen in several class demonstration this semester
mostly because it is very cold (-320 F).
Nitrogen was discovered in 1772 by Daniel Rutherford (a Scottish
botanist). Atmospheric nitrogen is relatively unreactive and
is sometimes used to replace air in packaged foods to preserve
freshness. You don't need to worry about details like this
for a quiz.
Oxygen is the second most abundant gas in the atmosphere.
Oxygen is the most abundant element (by mass) in the earth's
crust, in ocean water, and in the human body.
A couple of photographs of liquid oxygen are shown above.
It has a (very faint) pale blue color (I was pretty disappointed
when I saw the pictures for the first time because I had imagined
the liquid oxygen might be a deep vivid blue). I'd love to
bring some liquid oxygen to class but it's not readily available
on campus. And oxygen is very reactive. I suspect
you'd need to be very careful with liquid oxygen.
When heated (such as in an automobile
engine) the oxygen and nitrogen in air react to form compounds
such as nitric oxide (NO), nitrogen dioxide (NO2), and
nitrous oxide (N2O). Together as a group these
are called oxides of nitrogen; the first two are air pollutants,
the last is a greenhouse gas.
Here is a complete list of the 5 most abundant gases in
air. And a note about the figures
you find on these online notes, they may differ
somewhat from what was done in class. I often redraw them
after class, or use neater versions from a previous semester for
improved clarity.

Water vapor and argon are the 3rd and 4th
most abundant gases in the atmosphere. A 2% water vapor
concentration is listed above but it can vary from near 0% to as
high as 3% or 4%. Water vapor is, in many locations, the
3rd most abundant gas in air. In Tucson most of the year,
the air is dry enough that argon is in 3rd position and water
vapor is 4th.
Water vapor, a gas, is invisible. Water is the
only compound that exists naturally in solid, liquid, and
gaseous phases in the atmosphere.
Argon is an unreactive noble gas (helium, neon, krypton, xenon, and radon are also inert gases).
The concentration of carbon dioxide is much smaller than the
other gases (you don't need to remember the actual value).
That doesn't mean it isn't important. We'll spend a lot of
time this semester talking about water vapor and also carbon
dioxide. Water vapor and carbon dioxide are the two best
known and most important greenhouse gases. The greenhouse
effect warms the earth. Concentrations of greenhouse gases
such as carbon dioxide are increasing and there is concern this
will strengthen the greenhouse effect and cause global
warming. That's a topic we'll look at during the semester.
Here's a little more
explanation (from Wikipedia)
of why noble gases are so unreactive. Don't worry about
all these additional details. The noble gases have full
valence electron
shells. Valence electrons are
the outermost
electrons of an atom and are normally the only electrons that
participate in
chemical bonding. Atoms with full valence electron
shells are extremely stable and therefore do not tend to form
chemical bonds and have little tendency to gain or lose
electrons (take or give electrons to atoms of different
materials).
Noble gases are often used used in neon
signs; argon produces a blue color. The colors produced by
Argon (Ar), Helium (He), Kryton (Kr), Neon (Ne) and Xenon (Xe),
which are also noble gases, are shown above (source of the
images). The inert gases don't react with
the metal electrodes in the bulbs. Neons bulbs and
fluorescent bulbs often also contain mercury vapor (which means
you should dispose of them carefully when they burn out).
The mercury vapor emits ultraviolet light that strikes phosphors
of different kinds on the inside of the bulb. Different
colors are emitted depending on the particular type of phosphor
used in the bulb.
If we were using a textbook we'd
probably find something like the following table near the
beginning of the book (this table is from
a Wikipedia
article about the earth's atmosphere).
There is almost too much information in a chart like this, you
might be overwhelmed and not remember much. Also unless you
are familiar with the units on the numbers they might be
confusing. And notice you don't find water vapor in 3rd or
4th position near the top of the chart. That's because this
is a list of the gases in dry air. Unless you're
very attentive, you might miss that fact and might not see water
vapor down at the bottom of the chart.
Water plays an important role in the formation of clouds,
storms, and weather. Meteorologists are very interested in
knowing and keeping track of how much water vapor is in the air at
a particular place and time. One of the variables they use
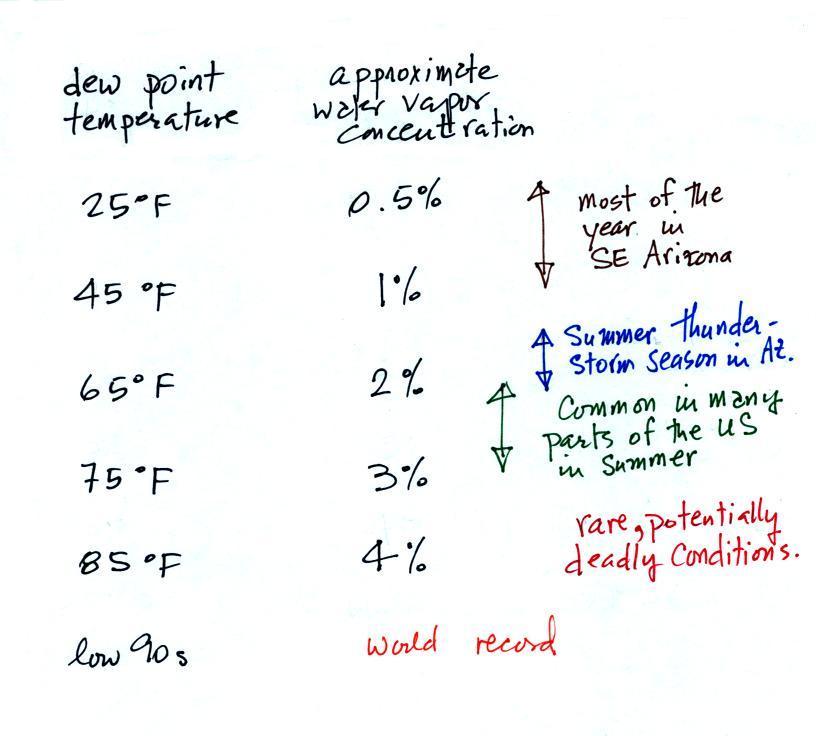
is the dew point temperature. The value of the
dew point gives you an idea of how much water vapor is actually in
the air. The higher the dew point value, the more water
vapor the higher the water vapor concentration.
The chart below gives a rough equivalence between dew point
temperature and percentage concentration of water vapor in the
air.
Air temperature will always be equal to or warmer than the
dew point temperature. Experiencing 80o dew points would be
very unpleasant and possibly life threatening because your
body might not be able to cool itself ( the air temperature
would probably be in the 90s or maybe even warmer).
Here's an
interesting link concerning unusually high, even record
setting dew point temperatures.
Click here
to see current dew point temperatures across the U.S.
At one time the dew point temperature was used to identify
the start of the summer monsoon season in Tucson (the summer
thunderstorm season). The following graph is
from the Tucson
National Weather Service Office.
Dates (running from June 1 through to the end of September) are
plotted along the x-axis and dew point temperature is shown on
the y-axis. Average daily dew point values observed this
year in blue. The red line shows typical daily average dew
point values for this time of the year.
Traditionally the summer monsoon would start when the daily
average dew point remained at or above 54 F (the green line
above) for 3 days in a row. That occurred on July 1 this
year.
Note how dew point values fell
into the low 40s for several days in early August. Dew
Points were near 70 F at the start of the week. That's
about has high as dew point and as humid as it ever gets in
Tucson.
Don't worry too much about all these dew point details.
Just remember that the higher the dew point temperature the more
water vapor is in the air and vice versa.
We can see liquid nitrogen but we can't see the nitrogen gas
being produced by the evaporation of liquid nitrogen. The
white cloud that surrounds the cup of liquid nitrogen isn't
nitrogen gas, what is it?
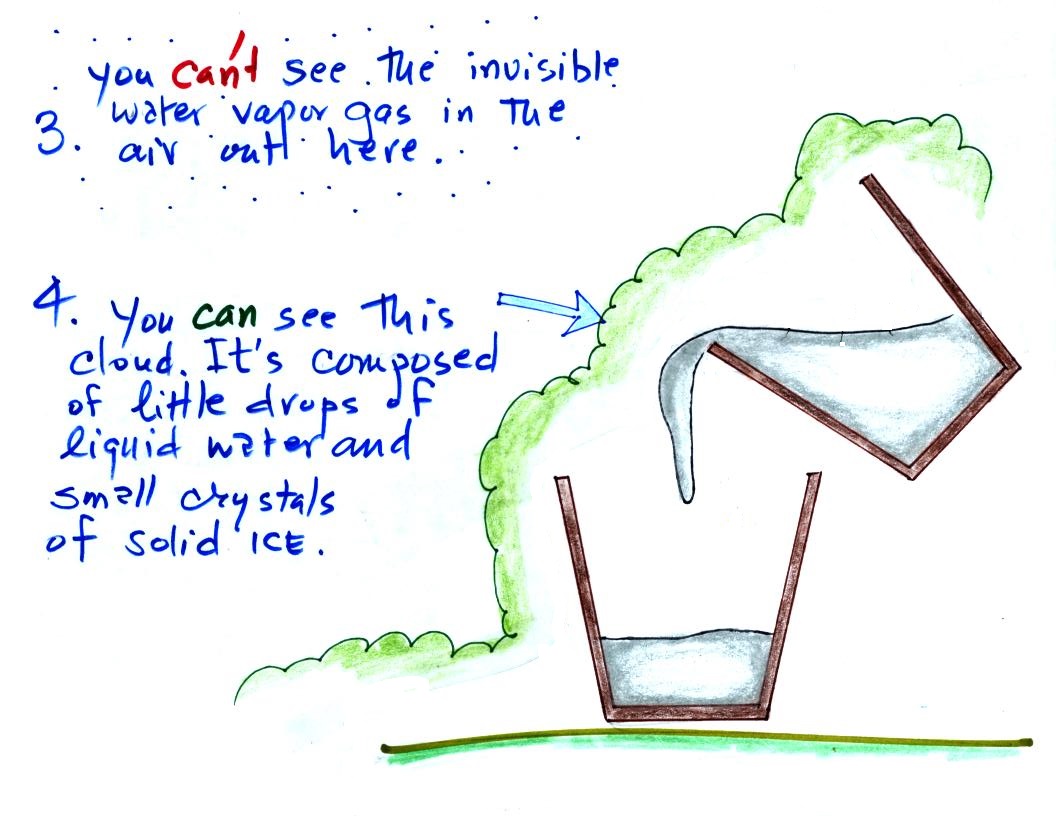
The white cloud isn't water vapor because water vapor, a gas,
is invisible just like nitrogen gas. When the air is cooled
however, by coming into contact with the liquid nitrogen, the
water vapor condenses and forms small droplets of water (liquid)
or ice crystals (solid). That's what you are
able to see, a cloud composed of water droplets or ice crystals.
This is where the dew point temperature's second job comes into
play.
If you cool air next to the ground to its dew point, water
vapor will condense and coat the ground with water. The
ground will be covered with dew. If a little thicker layer
of air is cooled fog will form.
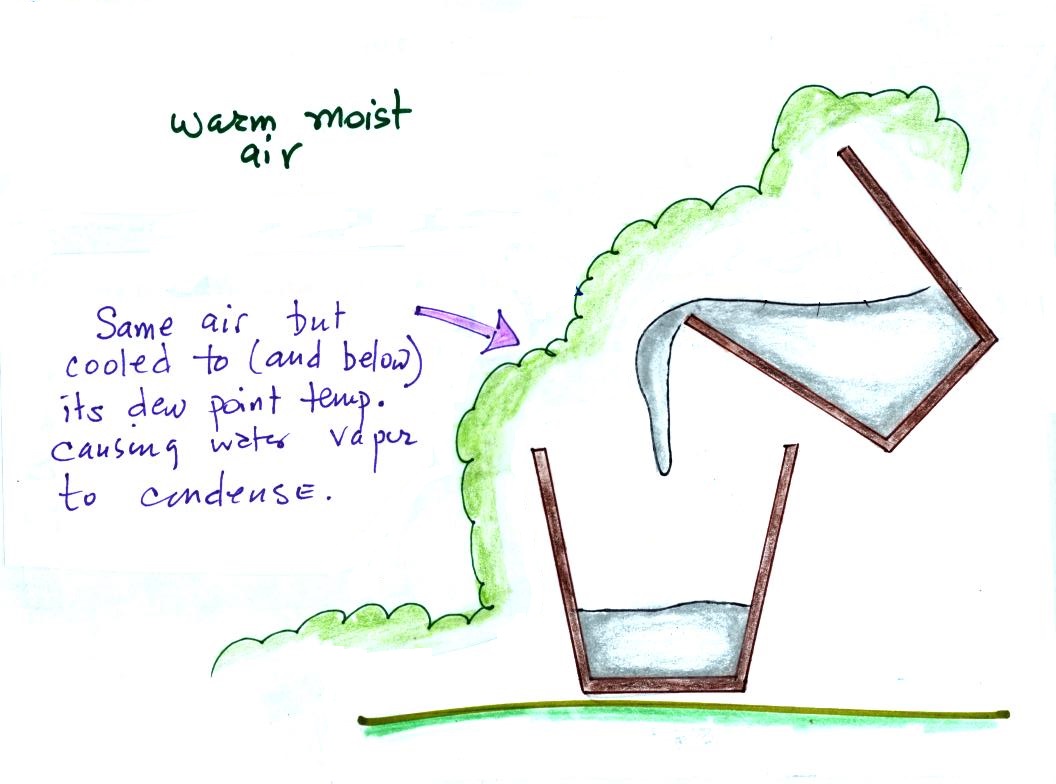
We can't ordinarily see the water vapor
(the moisture) in air. It's only when the moist air is
cooled to its dew point and the water vapor condenses that we
can see it.
We were almost out of time at this point. When you
leave class today, try to recall the 5 most abundant gases in
air without looking at your notes. Then try to remember
something about each of them. Here's what I'd recommend
Pluto's
Gate to Hell was discovered
earlier this year at the ancient
city of Hierapolis in southwestern
Turkey (Pluto was the Roman god of
the underworld, he was called
Hades by the Greeks)
The
picture above at left shows the
site as it appears now (source
of this photograph).
The gate is the opening in the
wall near the center of the
picture. The site as it
might have appear in ancient
times is shown above at
right. This photograph,
credited to Francesco
D'Andria, the lead Italian
archaelogist that announced
the discovery in March 2013,
is found in a news
report from the National
Geographic Society.
The
"gate" was built on top of a
cavern and, in ancient times,
a mist of deadly vapors could
be seen coming from the cave
(the mist is shown in the
right picture above).
Here's a quote from the Slate
article where I first
read about the discovery:
"Two
millennia ago, visitors to
Pluto's Gate could buy small
birds or other animals (the sale
of which supported the temple)
and test out the toxic air that
blew out of the mysterious
cavern. Only the priests,
high and hallucinating on the
fumes, could stand on the steps
by the opening to hell.
They would sometimes lead
sacrificial bulls inside, later
pulling out their dead bodies in
front of an awed crowd.
As
the Greek geographer,
philosopher, and prolific
traveler Strabo, who lived from
64/63 B.C. to 24 A.D., so
enticingly described it: 'This
space is full of a vapor so
misty and dense that one can
scarcely see the ground.
Any animal that passes inside
meets instant death. I
threw in sparrows and they
immediately breathed their last
and fell.' "
The Italian archaeologists working at the
site would occasionally notice birds dying if they flew into
the vapors coming from the came. The deadly gas was
carbon dioxide. Carbon dioxide is not ordinarily thought
of as a poisonous gas but in high enough concentrations it can
asphyxiate you (cause you to suffocate).