In order to describe the physics of these processes, we need some way to specify the water vapor content of the atmosphere. There are many ways to do this and we will use a few of them in this class. The first is something called vapor pressure.
Vapor pressure (e) is the gas pressure (force/area) exerted by water vapor molecules alone. The higher the concentration of water vapor molecules (number density), the higher the vapor pressure. The average air pressure at sea level is about 1013 millibars (mb). If the total air pressure is 1013 mb and water vapor makes up 1% of the air molecules, then the vapor pressure is 1% of 1013 mb or 10.13 mb. Water vapor is a trace gas in the atmosphere of Earth -- the maximum vapor pressure is never more than about 40 mb. For now the important concept is that vapor pressure is one way to keep track of the amount of the gas water vapor. For our purposes, the higher the vapor pressure, the greater the amount of water vapor in the air.
A Physical Explanation of the Processes of Evaporation and Condensation and the concept of Saturation, are provided in this figure depicting the relationship between vapor pressure and saturation. The figure shows what happens in a "closed system." The closed system does not allow air or water vapor from inside the flask to mix with air outside the flask. Evaporation and condensation are competing processes that occur simultaneously at the liquid water-air interface. The rate of evaporation can be defined as the number of water molecules that change phase from liquid to gas each second. The rate of evaporation is mainly set by the temperature of the liquid water. The higher the temperature of the liquid water, the faster the rate of evaporation. At the scale of individual molecules, a molecule of liquid water will evaporate (change phase from liquid to gas) whenever that molecule has enough energy to break free of the chemical bonds it has with neighboring liqiud water molecules. The higher the temperture of the liquid water, the higher the average energy of the liquid molecules, and the faster the rate of evaporation.
The rate of condensation can be defined as the number of water vapor molecules that change phase from gas to liquid each second. The rate of condensation depends mainly on the vapor pressure in the space above the liquid surface. The vapor pressure increases as the concentration of water vapor in the space above the liquid increases. Thus, the higher the concentration of water vapor molecules above the liquid surface, the faster the rate of condensation. Condensation occurs when a water vapor molecule collides with the liquid water surface, and chemically binds to the liquid water molecules. It should make sense that the higher the vapor pressure, the greater the rate of collisions, and the greater the rate of condensation.
Here is a brief explanation for the previously referenced figure depicting the relationship between vapor pressure and saturation. Panel (a) shows the system immediately after liquid water has been added with no time allowed for evaporation. The pressure gauge shows the pressure of the dry air inside the flask. The initial vapor pressure is zero. Consider the rates of evaporation and condensation. The rate of evaporation is set by the temperature, which is 20°C, while the rate of condensation is zero in panel (a) since the vapor pressure is zero. Since the rate of evaporation is greater than the rate of condensation, there will be a net movement of liquid water to water vapor. A short time later in panel (b), there is now a measureable vapor pressure. Keep in mind that the rate of evaporation has not changed since the temperature of the system is still 20°C, however the rate of condensation is increasing as the vapor pressure inside the flask increases. This continues until panel (c), when the vapor pressure has now increased to the point where the rate of evaporation is equal to the rate of condensation.
In a closed system, a dynamic equilibrium will be reached, where the rate of evaporation equals the rate of condensation. When this occurs, the the saturation vapor pressure (es ) can be measured, and the air inside the closed system is said to be saturated with water vapor. You may take saturation to mean the capacity or maximum amount of water vapor that will can exist in the air at a particular temperature. It is very important to understand that the higher the air temperature, the higher the saturation vapor pressure. Saturation vapor pressure has a strong temperature dependence. As the figure below shows, a small increase in temperature, results in a large increase in the saturation vapor pressure or capacity for water vapor in the air. In other words, warm air can hold more water vapor than cold air.
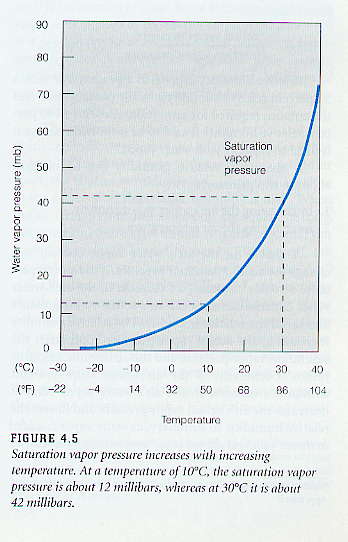
| 
|
The figure on the left shows the saturation vapor pressure for water for the range of temperatures typical at the surface of the Earth. Note from the figure caption that a temperature increase from 10°C to 30°C (50°F to 86°F), which can happen over the course of a day, causes the saturation vapor pressure to increase by a factor of three and a half, from 12 mb to 42 mb. Thus a small change in temperature results in a large change in the saturation vapor pressure or the maximum amount of water vapor that can exist in the air. It is important to understand that saturation vapor pressure (or the maximum amount of water vapor that can be in the air) increases exponentially as temperature increases.
The sharp increase in saturation vapor pressure with increasing temperature continues beyond 40°C (104°F). Water will boil when the saturation vapor pressure is equal to the surrounding air pressure. At sea level, water will boil when the saturation vapor pressure equals the sea level pressure, which has an average value of 1013 mb. As many of you know this happens when the water temperature is 100°C or 212°F. This is shown in the figure on the right side, which extends the temperature range up to 100°C. When water boils, bubbles of water vapor (gas form of water) are able form under the surface of the water and rise upward. The gas pressure inside these water vapor bubbles must be able to hold up against the surrounding pressure or they will be crushed. If the saturation vapor pressure is less than the surrounding air pressure, any bubble of water vapor that tries to form will be crushed by the surrounding air pressure. Therefore depending upon the exact value of the surrounding air pressure, water will begin to boil at a specific temperature, i.e., the temperature where the saturation vapor pressure is equal to the surrouding air pressure.
Near the Earth's surface, the vapor pressure is usually less than the saturation vapor pressure. (NOTE: the actual vapor pressure of the air can vary from zero up to the saturation vapor pressure). Therefore, the rate of evaporation is greater than the rate of condensation, i.e., liquid water near the Earth's surface is continually evaporating (changing phase from liquid to gas). You can easily convince yourself that this is true by leaving out a glass of water. All of the liquid will eventually evaporate. The reason the air near the ground does not reach saturation is because after the water evaporates, the water vapor is able to move away from the surface. It is not trapped as in the closed experiment described above.
Once water has evaporated it becomes part of the gases that make up the atmosphere. When air rises upward, it cools (the reason rising air cools will be explained later). As air cools, its saturation vapor pressure decreases, in other words, the maximum amount of water vapor that the air can hold decreases. If the air rises high enough and cools sufficiently, it will not be able to hold all the water vapor it contains. When this happens, water vapor must condense back to liquid water. This is how clouds form. Clouds are composed of tiny droplets of liquid water (and possibly ice). Water vapor is an invisible gas and cannot ever be seen. If you see it, e.g., clouds, steam, your breath on a cold day, it must be liquid water droplets. We will talk more about clouds soon.