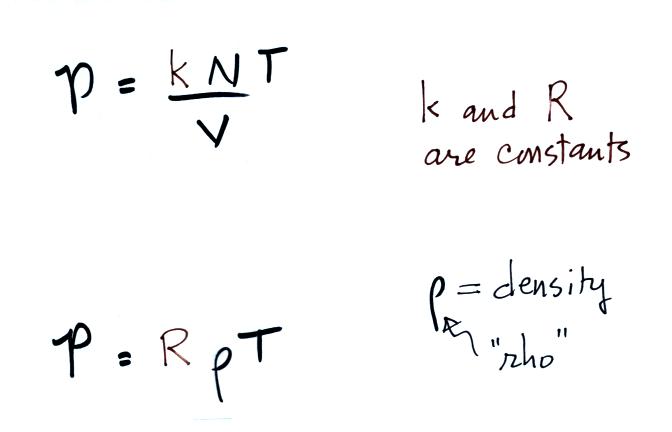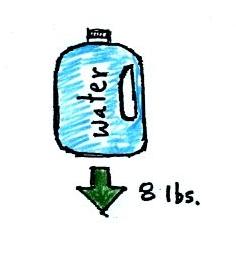Friday Feb. 5, 2016
Selections from Lucius
featured before class today: "Until We Get
There" (4:59), "Turn It
Around" (4:10), and "How Loud Your
Heart Gets" (6:58) and "Two
of Us on the Run" (4:51).
The Practice Quiz has been graded. This semester's
results is low but, as the chart below shows, is pretty
typical. The 68% average for the 11 am class is well above
average.
Semester
|
11 am MWF class
|
1 pm MWF class
|
S16
|
68%
|
61%
|
Semester
|
8 am T Th class
|
9:30 am T Th class
|
F15
|
61%
|
61%
|
S15
|
61%
|
61%
|
The 1S1P El Nino reports have also been graded and
were returned today.
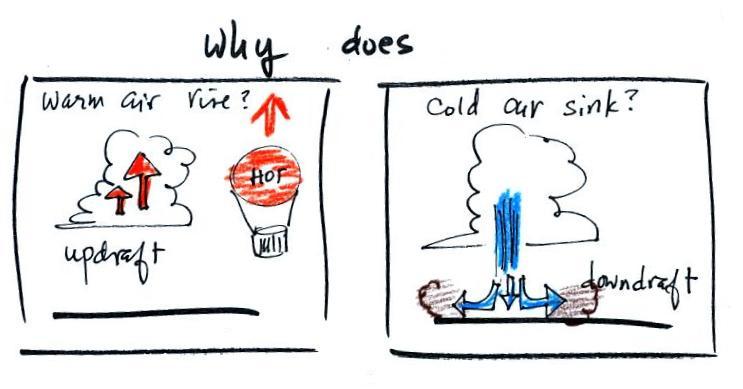
Back to trying to understand why warm air rises &
cold air sinks

|

|
Hot air balloons floating over the Rio
Grande river during the Albuquerque Balloon Fiesta (source
of the photo)
|
Photograph of a microburst, a localized
intense thunderstorm downdraft, that hit Wittmann Arizona
in July 2015. Surface winds of 55 MPH were measured.
(source
of the photo)
|
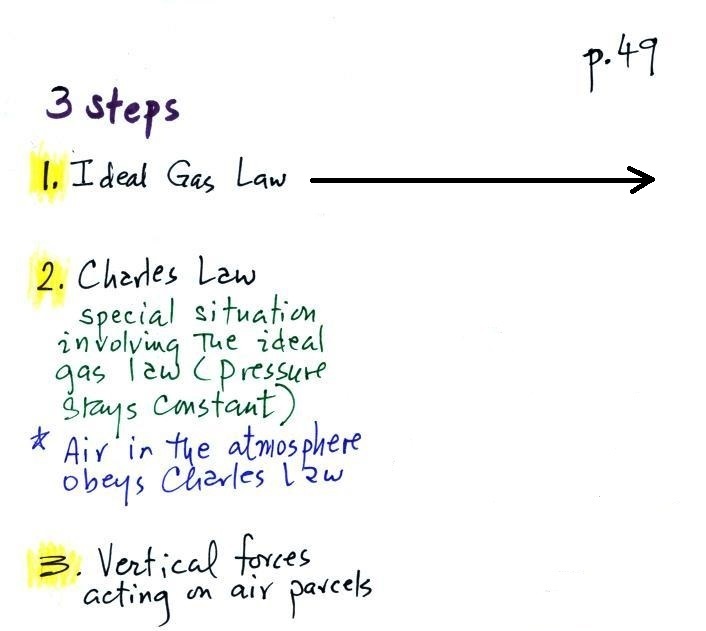
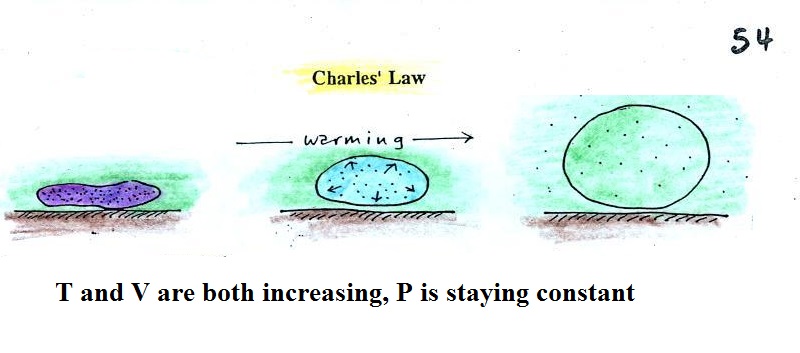
Step #2 Charles Law
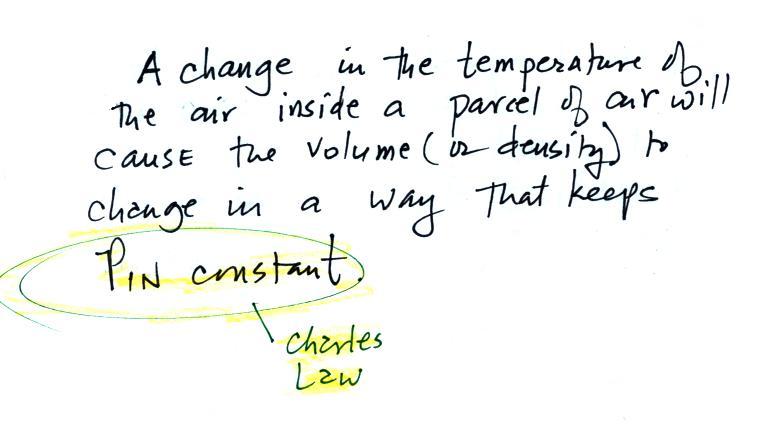
Charles Law means that P (pressure) in the ideal gas law
stays constant. Changing the temperature
of a volume of air will cause a change in density and volume;
pressure will stay constant. This is an important situation
because this is how volumes of air in the atmosphere behave.
This is probably the most difficult part of today's class and is
worked out in lots of detail.
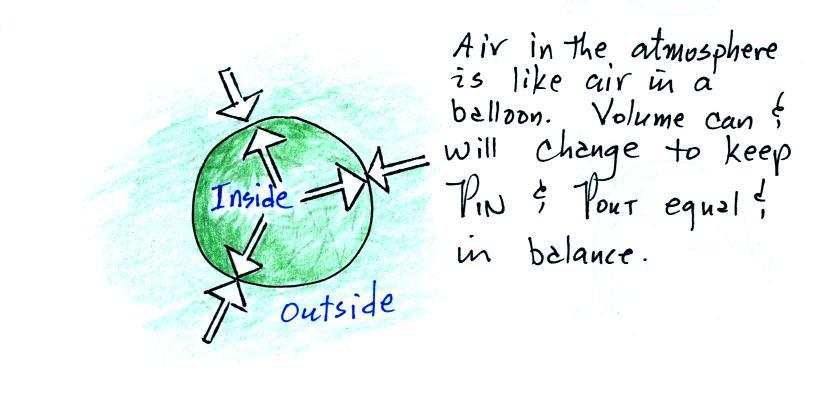
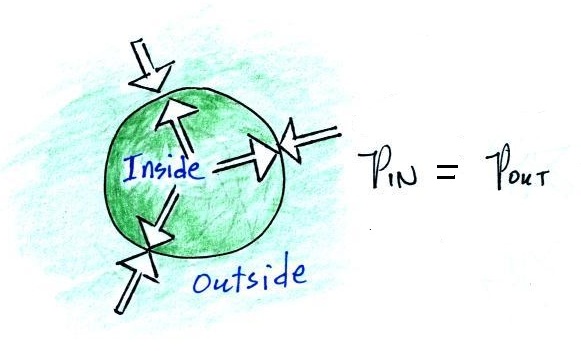
We will use a balloon of air. The air inside and
outside the balloon (or parcel) are exactly the same.
The pressure of the air surrounding the balloon pushing inward is
balanced by the pressure of the air inside the balloon that is
pushing outward. If we change something inside the balloon
that upsets this balance, the balloon would expand or shrink until
the pressures are again in balance.
Volumes of air in the atmosphere will behave the same
way.
First let's imagine warming the air inside a balloon. We'll
won't change the temperature of the air outside the balloon.
Increasing the temperature will momentarily increase the
pressure. This creates an imbalance. Now that P inside
is greater than P outside the balloon will expand.
Increasing the volume causes the pressure to start to
decrease. The balloon will keep expanding until P inside is
back in balance with P outside.
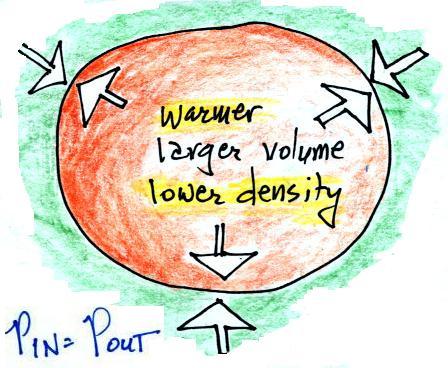
We're left with a balloon that is larger, warmer, and filled with
lower density air than it was originally.
The pressures inside and outside are again the same. The
pressure inside is back to what it was before we warmed the air in
the balloon. You can increase the temperature and volume of
a parcel together in a way that keeps pressure constant (which is
what Charles' law requires). This is equivalent to
increasing the temperature and decreasing the density together and
keeping the pressure constant.
In nature the change in temperature and volume occur
simultaneously. It's like jumping from the first to the last
step above.
Warming a parcel of
atmospheric air will cause the parcel volume to
increase, the density of the air in the parcel to
decrease, while pressure remains constant.
We can go through the same kind of reasoning and see what happens
if we cool the air in a parcel. Actually you should see if
you can figure it yourself. I've included all the steps
below. We'll just skip to the last step in class.
We'll start with a parcel of air that has the same temperature
and density as the air around it.
We'll cool the air inside the parcel. The air outside
stays the same.
Reducing the air temperature causes the pressure of the air
inside the balloon to momentarily decrease. Because the
outside air pressure is greater than the pressure inside the
balloon the parcel is compressed.
The balloon will get smaller and smaller (and the pressure
inside will get bigger and bigger) until the pressures inside and
outside the balloon are again equal. The pressure inside is
back to the value it had before you cooled the air in the parcel.
The first and last steps, without all the intermediate and
momentary details, are shown below.
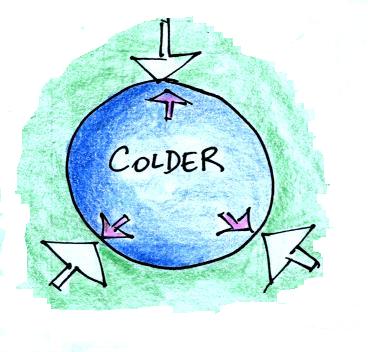
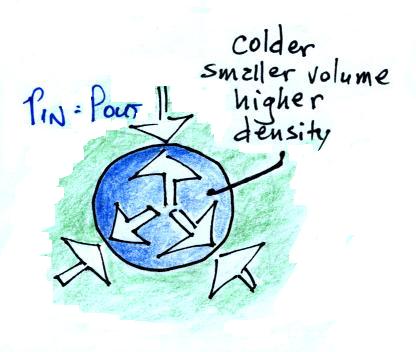
Cooling some air will cause volume to decrease and density to
increase while pressure stays constant.
If you want to skip all the details and just remember one
thing, here's what I'd recommend
Demonstration of Charles Law in action
Parcels of atmospheric air and air in balloons behave the same
way, they both obey Charles' Law. Charles Law
can be demonstrated by dipping a balloon in liquid nitrogen.
You'll find an explanation on the top of p. 54 in the photocopied
ClassNotes.
A balloon shrinks down to practically zero volume when dunked
in the liquid nitrogen. When pulled from the liquid nitrogen
the balloon is filled with very cold, very high density air.
Then the balloon starts to warm up.
The volume and temperature both increasing together in a way
that kept pressure constant (pressure inside the balloon is
staying equal to the air pressure outside the balloon).
Eventually the balloon ends up back at room temperature (unless it
pops while warming up).
All of that was just Step #2,
we've still got Step #3
Step #3 Two vertical forces acting on a parcel of air
in the atmosphere
(see p. 53 in the ClassNotes)
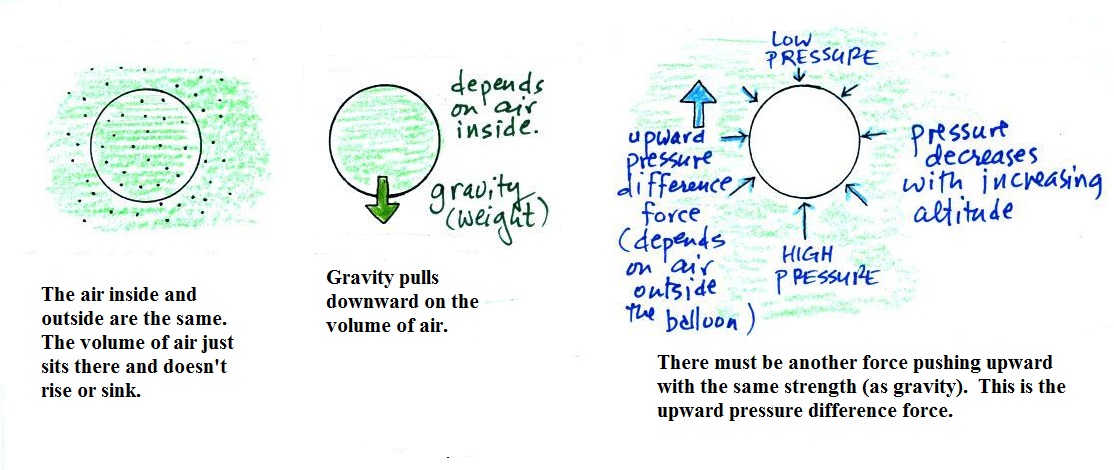
Basically it comes down to this - there
are two forces acting on a parcel of air in the atmosphere. They
are shown above.
The first force is gravity, it pulls downward. Most
everyone knows about this force. The strength of the
gravity force (the weight of the air in the parcel) depends on
the mass of the air inside
the parcel.
Second there is an upward pointing pressure difference
force. Not too many people know about this one. This
force is caused by the air outside (surrounding) the parcel.
Pressure decreases with increasing altitude. The pressure
of the air at the bottom of a parcel pushing upward is slightly
stronger than the pressure of the air at the top of the balloon
that is pushing downward. The overall effect is an upward
pointing force.
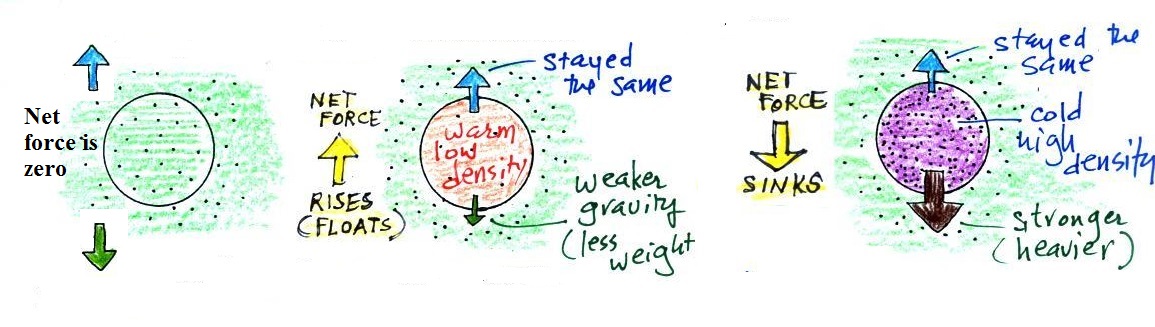
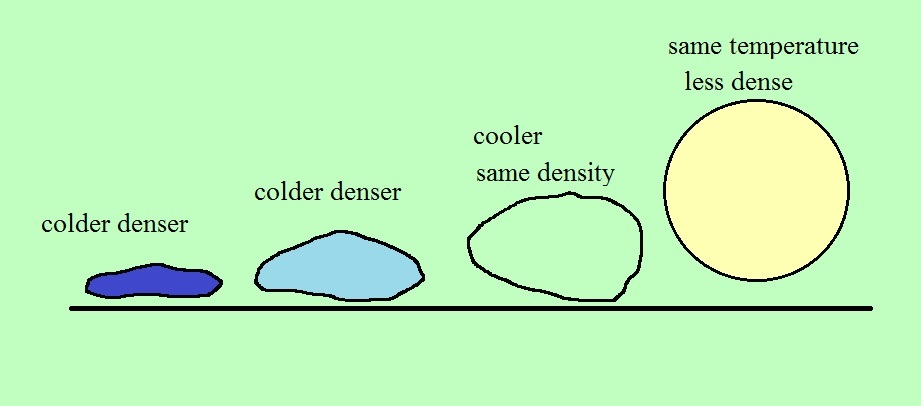
When the air inside a parcel is exactly the same as the air
outside (same densities), the two forces are equal in strength
and cancel out. The parcel is neutrally buoyant and it
wouldn't rise or sink, it would just hover.
We'll replace the air inside the balloon
with either warm low density air or cold high density
air.
In the first case, a balloon with warm low density air won't
weigh as much. The gravity force is weaker. The
upward pressure difference force doesn't change (because it is
determined by the air outside the balloon which hasn't changed)
and ends up stronger than the gravity force. The balloon
will rise.
Conversely if the air inside is cold high density air, it
weighs more. Gravity is stronger than the upward pressure
difference force and the balloon sinks.
It all comes down to a question of how the density of
the air in a parcel compares to the density of the air
surrounding the parcel. If the parcel is filled
with low density air it will rise. A parcel full of high
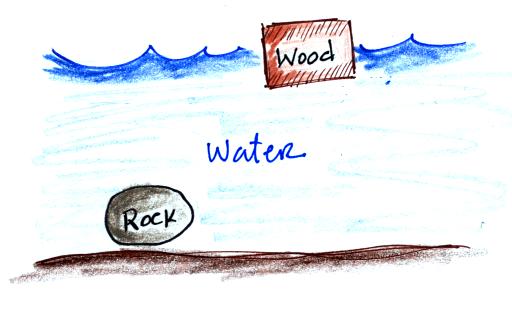
density air will sink. That's true of things other than
air. Wood floats in water because it is less dense than
water.
We'll look at this in a different way after the demonstration
Here's a short demonstration of the role
that density plays in determining whether a balloon will rise or
sink (or hover)
Convection demonstration
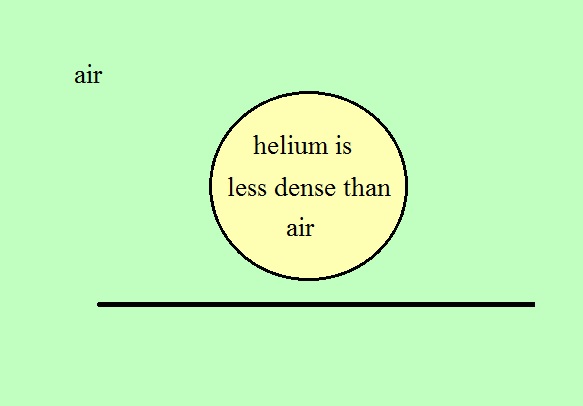
We used balloons filled with helium (see
bottom of p. 54 in the photocopied Class Notes).
Helium is less dense than air even when it has the same
temperature as the surrounding air. The downward
gravity force (weight of the helium filled balloon) is
weaker than the upward pressure difference force. You
don't need to warm a helium-filled balloon to make it rise.
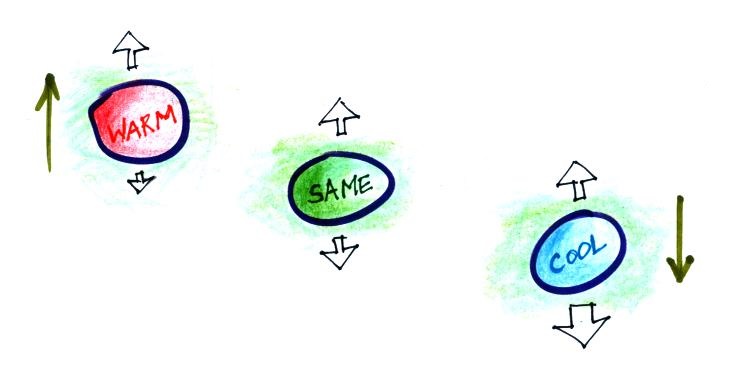
We dunk the helium filled balloon in
liquid nitrogen to cool it off. When you pull the balloon
out of the liquid nitrogen it has shrunk. The helium is
denser than the surrounding air. I set it on the table (dark
blue above) and it just sat there.
As the balloon of helium warms and expands its density decreases
(light blue). For a brief moment it has the same density as
the surrounding air (green). It's neutrally buoyant at this
point. Then it warms back to near room temperature where it
is again less dense than the air and lifts off the table (yellow).
Free convection
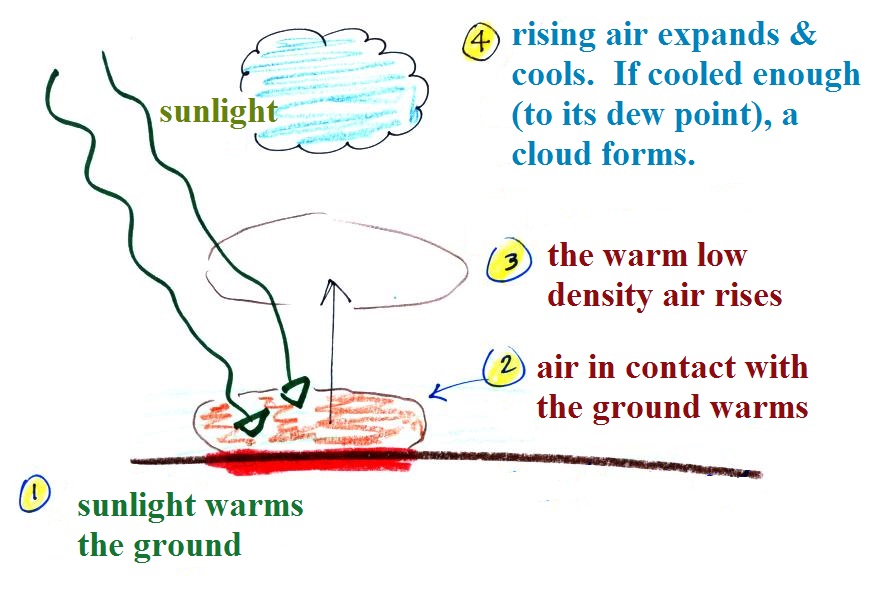
Something like this happens in the atmosphere.
Sunlight shines through the atmosphere. Once it reaches the
ground at (1) it is absorbed and warms the ground. This in
turns warms air in contact with the ground (2) As this air
warms, its density starts to decrease (pressure is staying
constant). When the density of the warm air is low enough,
small "blobs" of air separate from the air layer at the ground and
begin to rise, these are called "thermals." (3) Rising air
expands and cools (we've haven't covered this yet and it might
sound a little contradictory). If it cools enough (to the
dew point) a cloud will become visible as shown at Point 4.
This whole process is called convection; many of our summer
thunderstorms start this way.
Archimedes' principle
Here's another way of trying to understand why warm air
rises and cold air sinks - Archimedes Law or Principle (see pps
54a & 54b in the ClassNotes). It's perhaps a
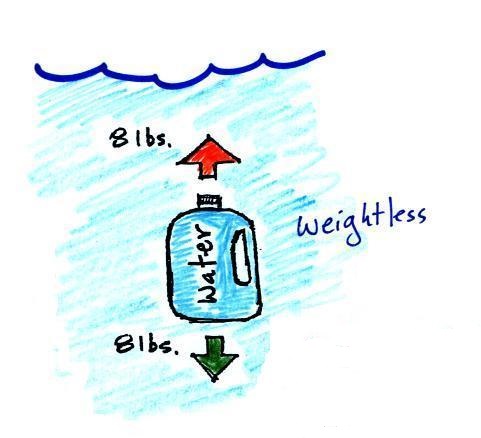
simpler way of understanding the topic. A bottle of water
can help you to visualize the law.
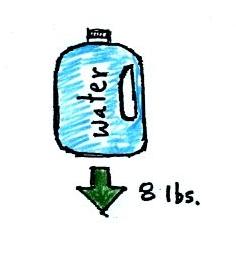
A gallon of water weighs about 8
pounds (lbs). I wouldn't want to carry that much water on
a hike unless I really thought I would really need it.
Here's something that is kind of surprising.
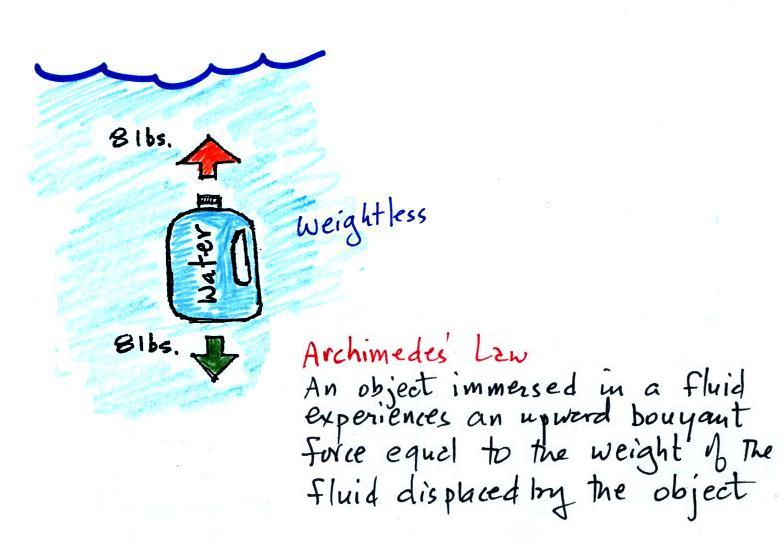
If you submerge the gallon of water in a swimming pool, the
jug becomes, for all intents and purposes, weightless. The
weight of the water (the downward gravity force) doesn't just go
away. Once the jug is immersed, an upward force appears
and it is strong enough to cancel out gravity. Archimedes'
recognized that this would happen and was able to determine how
strong the upward force would be.
In this case the 1 gallon bottle will displace 1 gallon of
pool water. One gallon of pool water weighs 8
pounds. The upward buoyant force will be 8 pounds, the
same as the downward force. The two forces are equal
and opposite.
What Archimedes law doesn't really tell you is what
causes the upward buoyant force. You should know what
the force is - it's the upward pressure difference force.
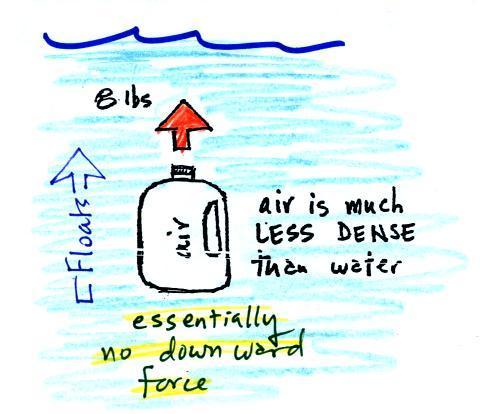
|
|
| We've poured out the water and filled
the 1 gallon jug with air. Air is much less dense
than water; compared to water, the jug will weigh
practically nothing. But it still displaces a gallon
of water and experiences the 8 lb. upward buoyant
force. The bottle of air would rise (actually it
shoots up to the top of the pool). |
The density of the material inside and
outside the bottle are the same. A bottle
filled with water is weightless. |
Next we'll fill the bottle with something
denser than water (I wish I had a gallon of mercury)
|
 |
| Sand is about 50% denser than
water. The weight of a gallon of sand is more
than a gallon of water. The downward force is
greater than the upward force and the bottle of sand
sinks. |
|
You can sum all of this up by saying anything that is
less dense than water will float in water, anything that is
more dense than water will sink in water.
Most types of wood will float (ebony and
ironwood will sink). Most rocks sink (pumice is an
exception).
The fluid an
object is immersed in doesn't have to be water, or a
liquid for that matter. You could immerse an
object in air. So we can apply Archimedes Law to
parcels of atmospheric air.
Air that is less dense (warmer) than the air around it will
rise. Air that is more dense (colder) than the air around
it will sink.
Here's a little more
information about Archimedes that I didn't mention in class.
I wanted to show one last application of
some of what we have been learning - a Galileo
thermometer. It's a new acquisition of mine and fairly
fragile.
The left figure above comes from an
interesting and informative article in Wikipedia. The
right figure is a closeup view of the thermometer I brought to
class.
Here's an explanation of how the thermometers work. We didn't cover this in class.
This is not something you need to worry about but I included it
just in case you're curious.
The fluid in the thermometer will expand slightly if it
warms. It will shrink when it cools.
The changes in the volume of the fluid will change the fluid's
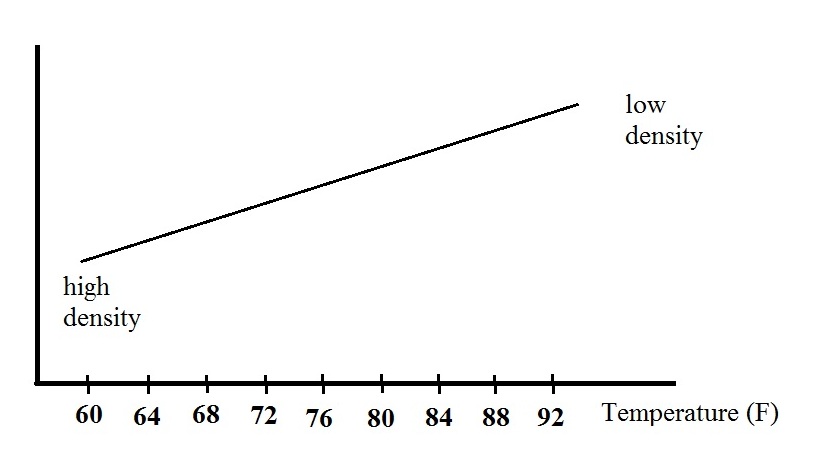
density. The graph above shows how the fluid density might
change depending on temperature. Note lower densities are
found near the top of the graph.
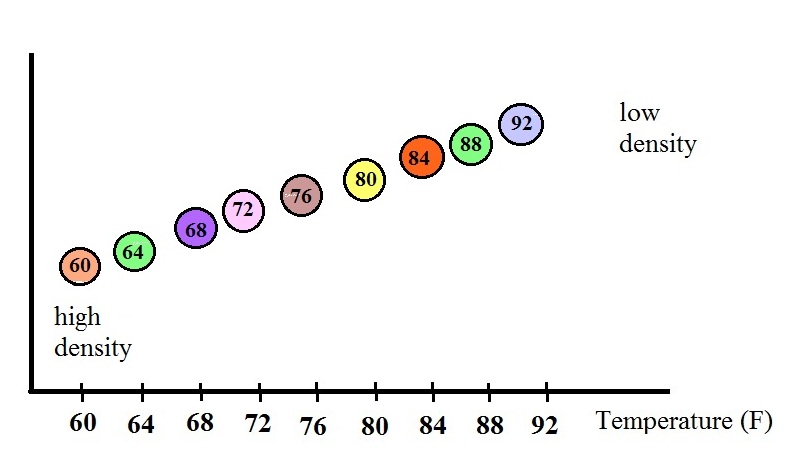
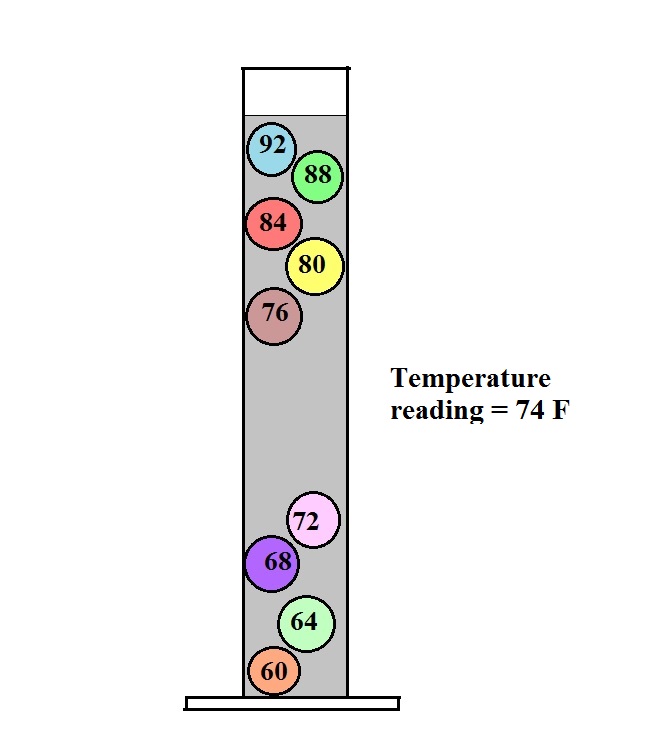
The colored balls in the thermometer all
have slightly different densities. They also have little
temperature tags. The 60 F ball has a density equal to the
density of the fluid at 60 F. The 64 F ball has a
slightly lower density, the density of the fluid when it has
warmed to 64 , and so on. The densities of the floats don't
change.
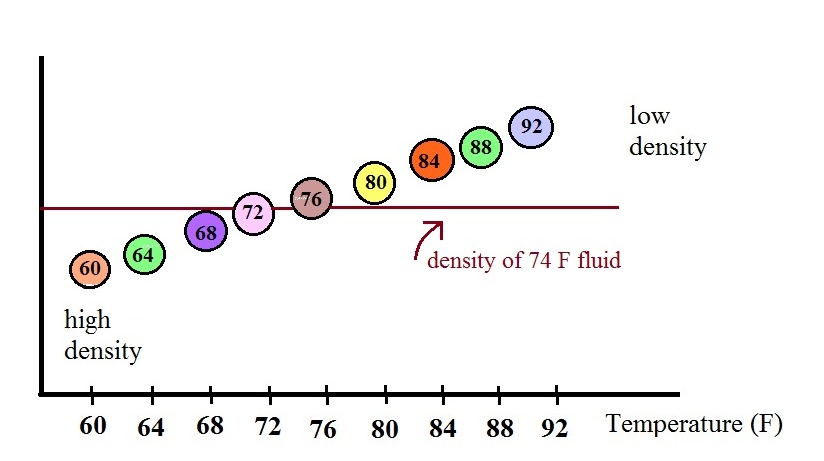
In use the density of the fluid in the thermometer will
change depending on the temperature. The densities of the
balls remain constant. As an example we will that the fluid
in the thermometer has a temperature of 74 F. The 60, 64,
68, and 72 F balls will all have densities higher than the fluid
(they lie below the 74F line in the graph above) and will
sink. The remaining balls have densities lower than the
fluid and will float.
The lower most floating ball in the illustration has a 76 F
temperature tag. The uppermost of the balls that have sunk
reads 72 F. The temperature is something between 72 F and 76
F. With this thermometer you can only determine temperature
to the nearest 4 F. Also the thermometer takes quite a while
(maybe an hour or two) to respond to a change in temperature.