Tuesday Oct. 2, 2007
The 2nd Optional Assignment was collected in class today.
I forgot to hand out the 3rd Optional Assignment (some people picked it
up at the end of the period). It isn't due until next Tuesday so
you can pick up a copy in class on Thursday. Copies are also
available in PAS 588.
The Experiment #2 reports and the revised
Expt. #1 reports are due next Tuesday. Expt. #2 doesn't take long
to perform. Collect your data, return your materials this week,
and pick up the supplementary information sheet.
We are making pretty good progress
on grading the 1S1P reports. Some of the reports were
returned in class today
We quickly
reviewed the discussion of electromagnetic radiation found at the end
of last Thursday's notes.
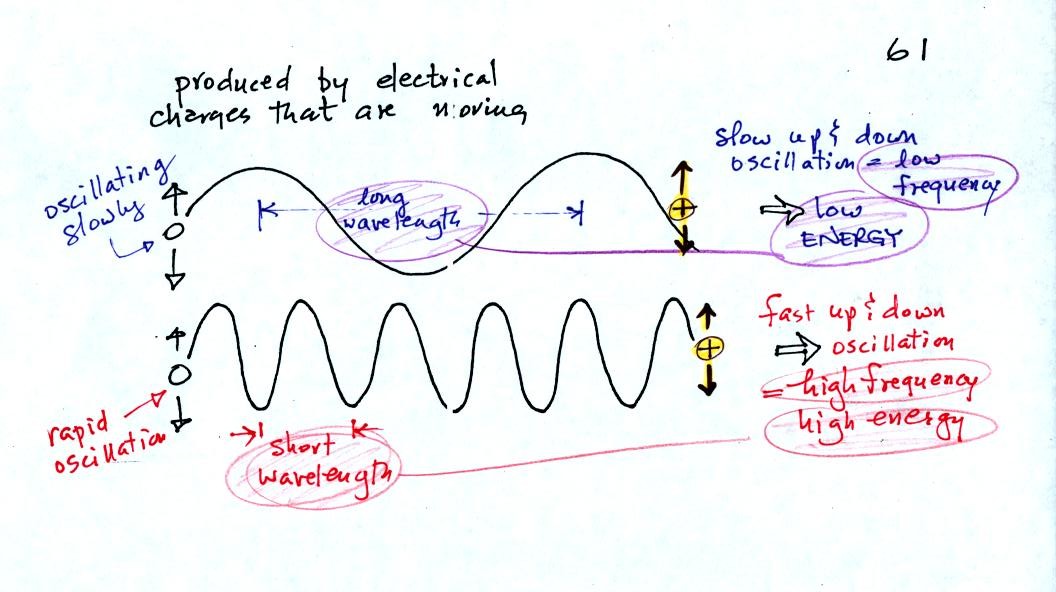
EM radiation is created when you cause a charge to move up
and down.
If you move a charge up and down slowly (upper left in the
figure above) you would produce long wavelength radiation that would
propagate out to the right at the speed of light. If you move the
charge up and down more rapidly you produce short wavelength radiation
that propagates at the same speed.
Once the EM radiation encounters the charges at the right side of the
figure above the EM radiation causes those charges to oscillate up and
down. In the case of the long wavelength radiation the charge at
right oscillates slowly. This is low frequency and low energy
motion. The short wavelength causes the charge at right to
oscillate more rapidly - high frequency and high energy.
The characteristics long wavelength - low frequency - low energy go
together. So do short wavelength - high frequency - high energy.
Note that the two different types of radiation both propagate at the
same speed.
The figure above starts to give you an idea of how energy is
transported from one place to another by electromagnetic radiation.
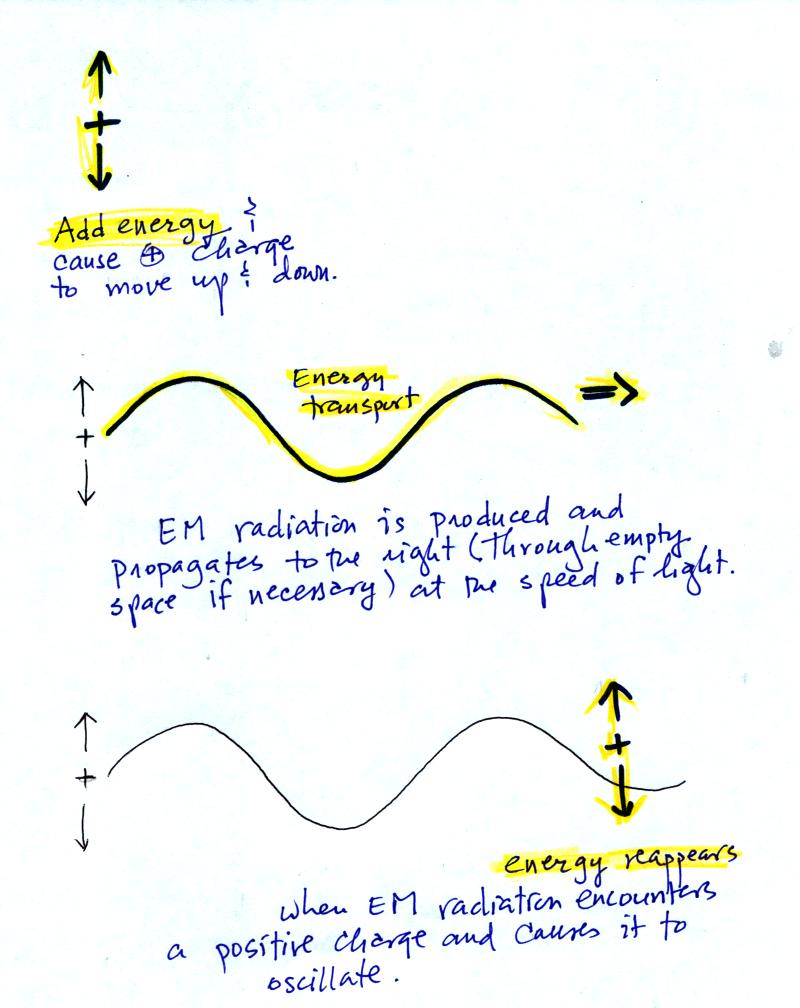
You add energy when you cause an electrical charge to move up and
down
and create the EM radiation (top).
In the middle figure, the EM
radiation then travels out
to the
right (it could be through empty space or through something like the
atmosphere).
Once
the EM radiation encounters an electrical charge at another location
(bottom right),
the energy reappears as the radiation causes the charge to move.
Energy
has been transported from left to right.
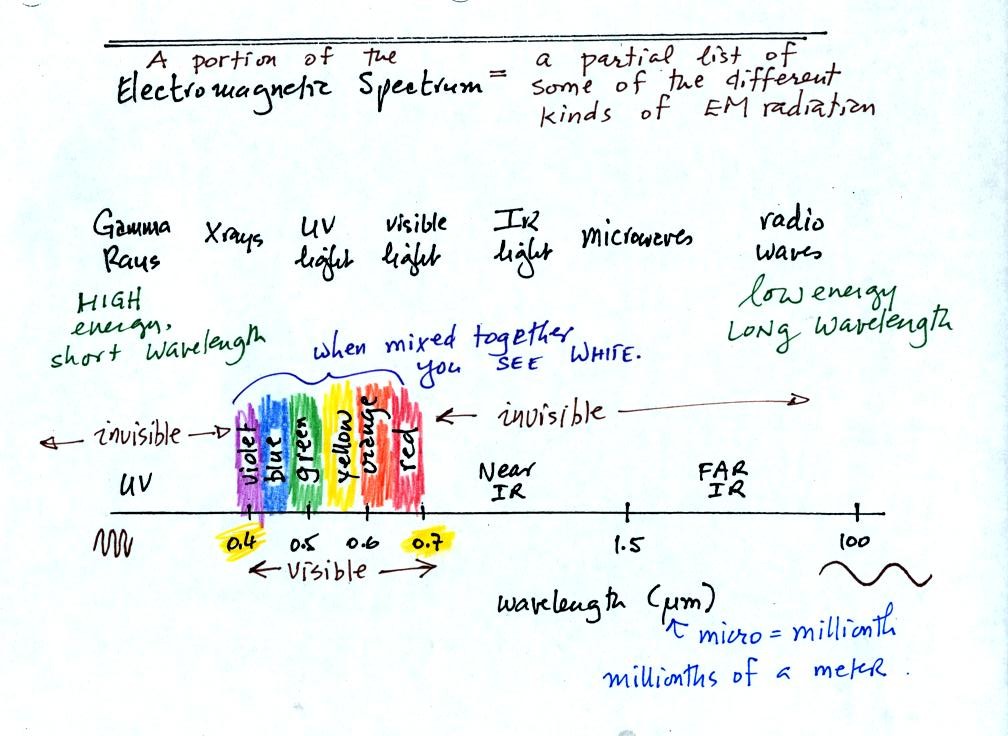 This is really just a partial
list of some of the different
types of EM
radiation. In the top list, shortwave length and high energy
forms of EM radiation are on the left (gamma rays and X-rays for
example). Microwaves and radiowaves are longer wavelength, lower
energy forms of EM radiation.
This is really just a partial
list of some of the different
types of EM
radiation. In the top list, shortwave length and high energy
forms of EM radiation are on the left (gamma rays and X-rays for
example). Microwaves and radiowaves are longer wavelength, lower
energy forms of EM radiation.
We will mostly be concerned with just ultraviolet light (UV), visible
light (VIS), and infrared light (IR). Note the micrometer
(millionths of a meter) units used for wavelength. The visible
portion of the spectrum falls between 0.4 and 0.7 micrometers (UV and
IR light are both invisible). All of the vivid colors shown above
are just EM radiation with slightly different wavelengths. WHen
you see all of these colors mixed together, you see white light.

1.
Unless an object is very cold (0
K) it will emit EM
radiation. All the people, the furniture, the walls and the floor
in the classroom are emitting EM radiation. Often this radiation
will be invisible so that we can't see it and weak enough that we can't
feel it. Both the amount and kind (wavelength) of the emitted
radiation depend on the object's temperature.
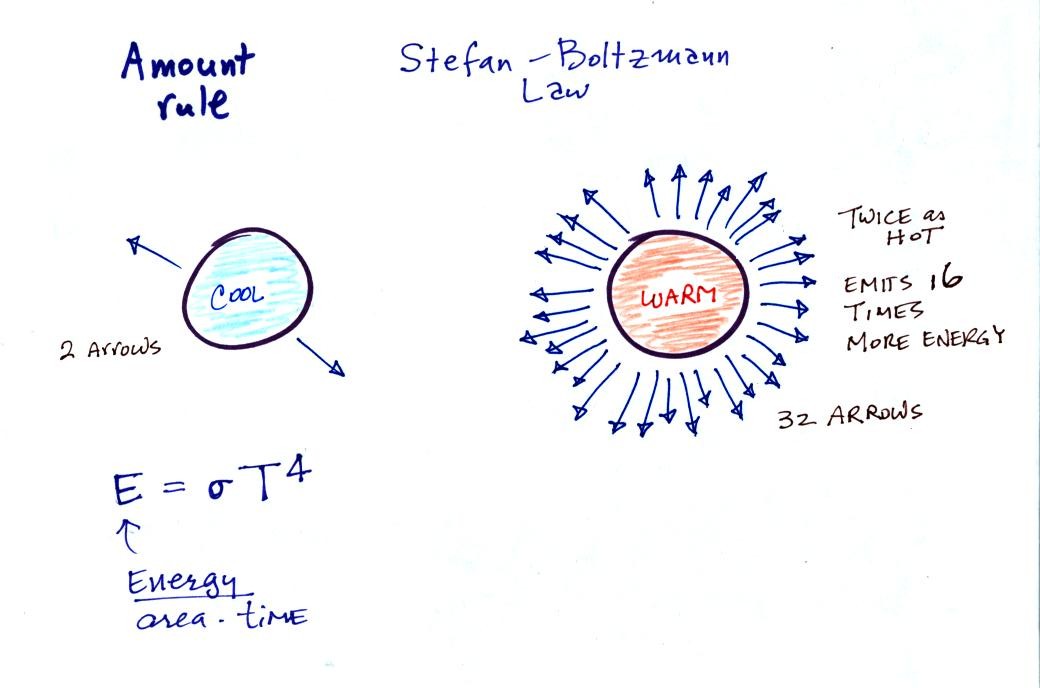
2.
The second rule allows you to
determine the amount of EM radiation (radiant energy) an object will
emit. Don't worry about the units,
you can think of this as amount, or rate, or intensity.
Don't worry about σ either, it is just a
constant. The amount depends on temperature to
the fourth
power. If the temperature of an object doubles the amount of
energy emitted will increase by a factor of 2 to the 4th power
(that's 2 x 2 x 2 x 2 = 16). A hot object just doesn't emit a
little more energy than a
cold object it emits a lot more energy than a cold object. This
is illustrated in the following figure:
3.
The third rule tells you something
about the kind of radiation emitted
by an object. We will see that objects usually emit radiation at
many different wavelengths. There is one wavelength however at
which the object emits more energy than at any other wavelength.
This is called lambda max (lambda is the greek character used to
represent wavelength) and is called the wavelength of maximum
emission. The third rule allows
you to calculate "lambda max." This is illustrated below:

The
following graphs (at the bottom of p. 65 in the photocopied Class
Notes) also help to
illustrate the Stefan-Boltzmann law
and Wien's law.
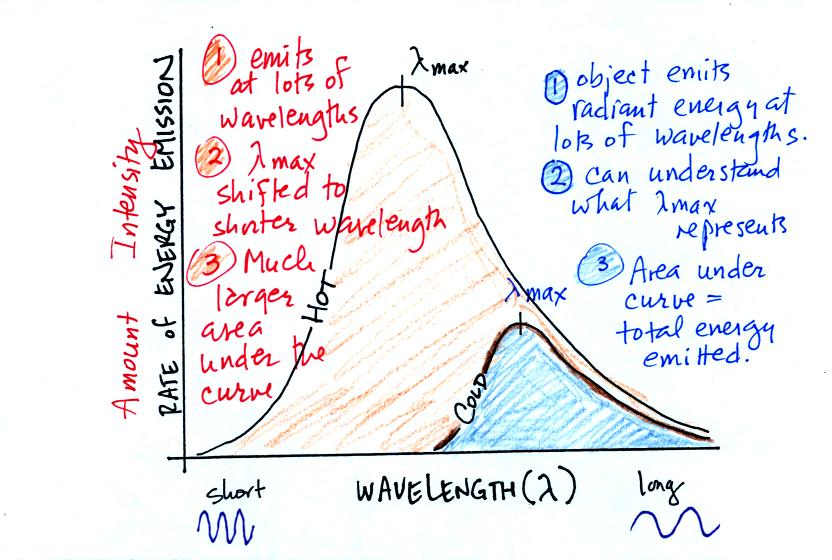
Notice first that both and warm and the cold objects emit radiation
over a range of wavelengths.
The area under the warm object curve is much bigger than the area under
the cold object curve. The area under the curve is a measure of
the total radiant energy emitted by the object. This illustrates
the fact that the warmer object emits a lot more radiant energy than
the colder object.
Lambda max has shifted toward shorter wavelengths for the warmer
object. This is Wien's law in action. The warmer object is
emitting a lot of short wavelength radiation that the colder object
doesn't emit.
A bulb connected
to a dimmer switch can be used to demonstrate the
rules above. We'll be interested in and looking at the EM
radiation emitted by
the tungsten filament in the bulb.
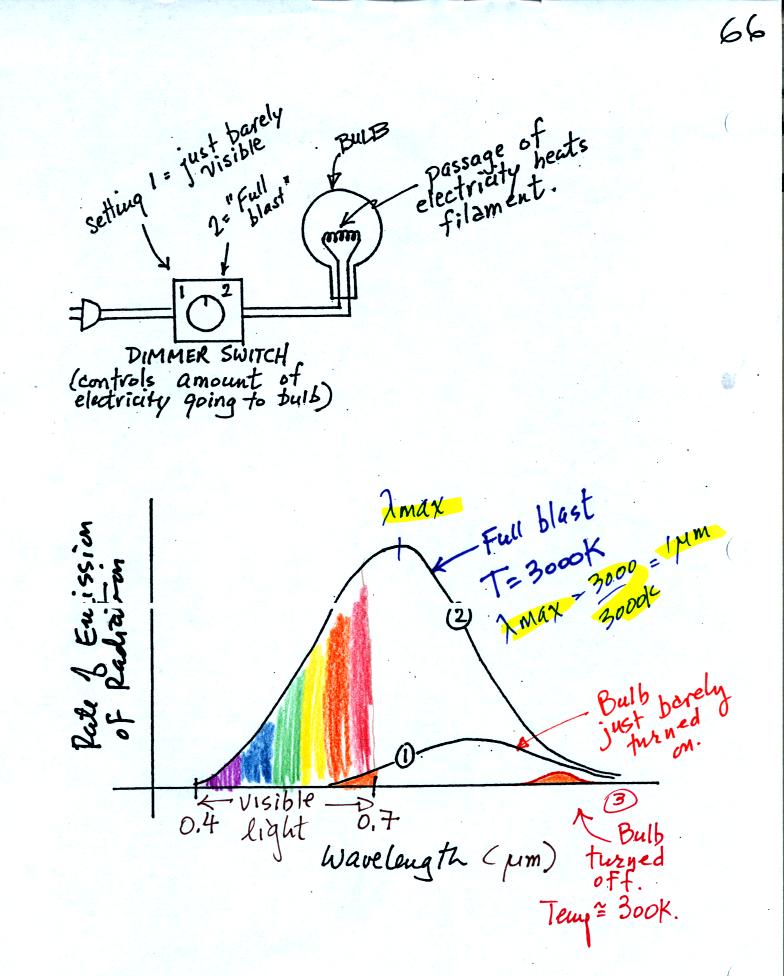
We start with the bulb turned off (Point 3 near the bottom right part
of the figure above). The
filament will be at room temperature which we will assume is around 300
K (remember that is a reasonable and easy to remember value for the
average temperature of the earth's surface). The bulb will be
emitting radiation. The radiation is very weak so we
can't
feel it. It is also long wavelength far IR radiation so we
can't see it. The wavelength of peak emission is 10 micrometers.
Next we use the dimmer switch to just barely turn the bulb on (the
temperature of the filament is now about 900 K).
The bulb wasn't very bright at all and had an orange color. This
is curve 1 in the figure. Note the far left end of the curve has
moved left of the 0.7 micrometer mark - into the visible portion of the
spectrum. That is what you are able to see the small fraction of
the radiation emitted by the bulb that is visible light (but just
long wavelength red and orange light). Most of the radiation
emitted by the bulb is to the right of the 0.7 micrometer mark and is
invisible IR radiation (it is strong enough now that you could feel it
if you put your hand next to the bulb).
Finally we turn on the bulb completely. The filament temperature
is now about 3000K. The bulb is emitting a lot more visible
light, all the colors, though not all in equal amounts. The bulb
was also much brighter (a 200 Watt bulb was used so it was pretty
bright). The mixture of the colors produces a warm
white light. It is warm because it is a mixture that contains a
lot more red, orange, and yellow than blue, green, and violet
light.
Small diffraction gratings were handed out in class. A
diffraction grating will spread out all of the colors in the white
light emitted by the bulb. You could see the vivid violets, blue,
green, yellow, orange, and red light that make up white light.
It is interesting that most of the radiation emitted by
the bulb is still in the IR portion of the spectrum (lambda max is 1
micrometer). This is
something I forgot to mention in class. This is
invisible light. A tungsten bulb like this is not especially
efficient, at least not as a source of visible light.
The sun
emits electromagnetic radiation. That shouldn't come as a surprise
since you can see it and feel it. The earth also emits
electromagnetic radiation. It is much weaker and invisible.
The kind and amount of EM radiation emitted by the earth and sun depend
on their respective temperatures.
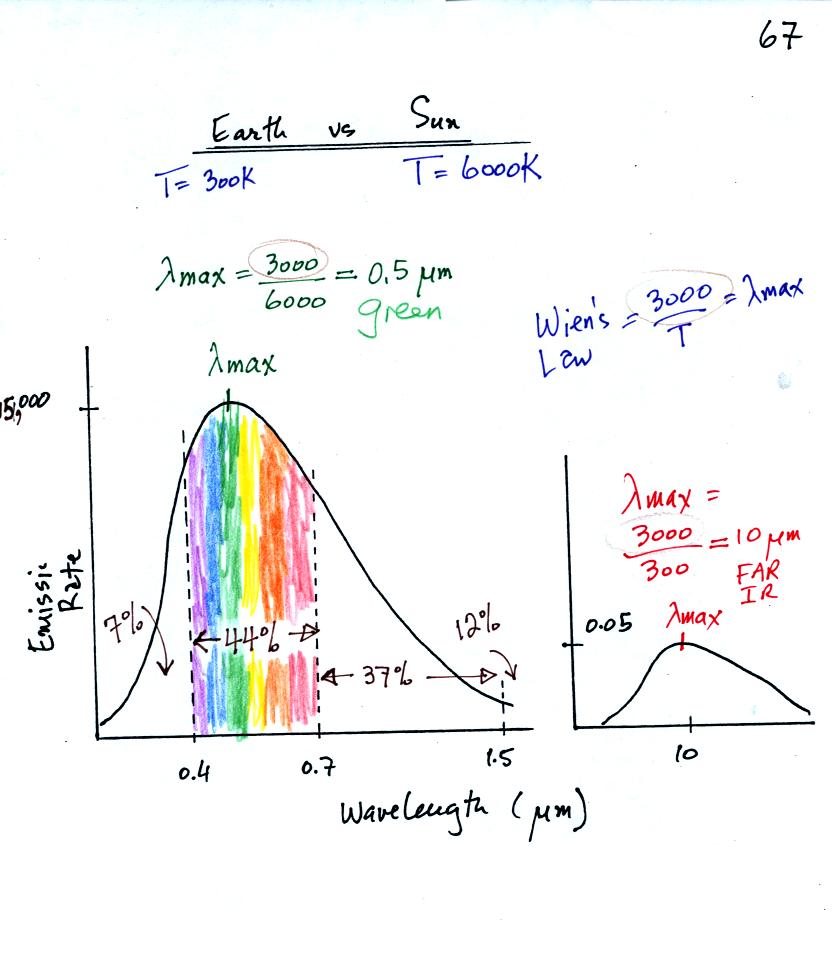
The curve on the left is for the sun. We first used
Wien's
law and a temperature of 6000 K to calculate lambda max and got
0.5 micrometers. This is green light; the sun emits more green
light than any other kind of
light. The sun doesn't appear green because it is also emitting
lesser amounts of violet, blue, yellow, orange, and red - together this
mix of
colors appears white. 44% of the radiation emitted by the sun is
visible light, 49% is IR light (37% near IR + 12% far IR), and 7%
is ultraviolet light. More than half of the light emitted by the
sun is invisible.
100% of the light emitted by the earth (temperature = 300 K) is
invisible IR light. The
wavelength of peak emission for the earth is 10 micrometers.
Because the sun (surface of the
sun) is 20 times hotter than the earth a square foot of the sun's
surface emits energy at a rate that is 160,000 times higher than a
square foot on the
earth. Note
the vertical scale on the earth curve is different than on the sun
graph. If both the earth and sun were plotted with the same
vertical scale, the earth curve would be too small to be seen.
We now
have most of the tools we will need to begin to study energy balance on
the earth. It will be a balance between incoming sunlight
energy and outgoing energy emitted by the earth. We will look at
the simplest case, first, the earth without an atmosphere (or at least
an atmosphere without greenhouse gases)
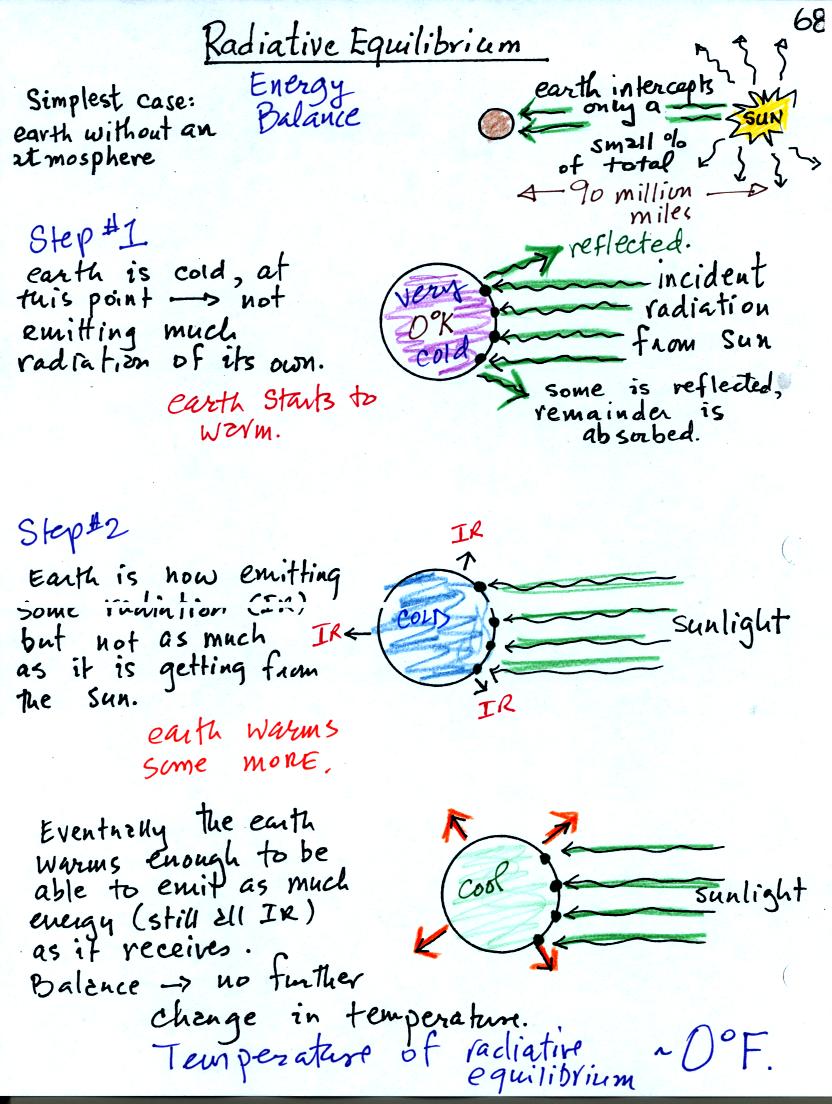
You might first wonder how, with the sun emitting so much more
energy than the earth, it is possible for the earth to be in energy
balance with the sun (without the earth getting really hot like the
sun). The earth is located about 90 million miles
from the sun and therefore only absorbs a very small fraction of the
energy emitted by the sun.
To understand how energy balance occurs we start, in Step #1, by
imagining that the earth starts out very cold and is
not emitting
any EM radiation at all. It is absorbing sunlight however so it
will
begin to warm.
Once the earth starts to warm it will also begin to emit EM
radiation, though not as much as it is getting from the sun (the
slightly warmer earth in the middle picture is now colored blue).
Because the earth is still gaining more energy than it is losing the
earth will warm some more.
Eventually it will warm enough that the earth (now shaded green) will
emit the same amount
of energy (though not the same wavelength energy) as it absorbs from
the sun. This is radiative equilibrium, energy balance. The
temperature at
which this occurs is 0 F.
That is called the temperature of radiative equilibrium. You
might remember this is the figure for global annual average surface
temperature on the earth without the greenhouse effect.
Next we
are going to see that the atmosphere behaves somewhat differently, when
it comes to emitting and absorbing electromagnetic radiation, than the
earth or the sun. We will need to understand, in particular, how
the atmosphere filters incoming sunlight and outgoing IR radiation
emitted by the ground. Then we will be able to study energy
balance on the earth with an atmosphere (and one that contains
greenhouse gases).
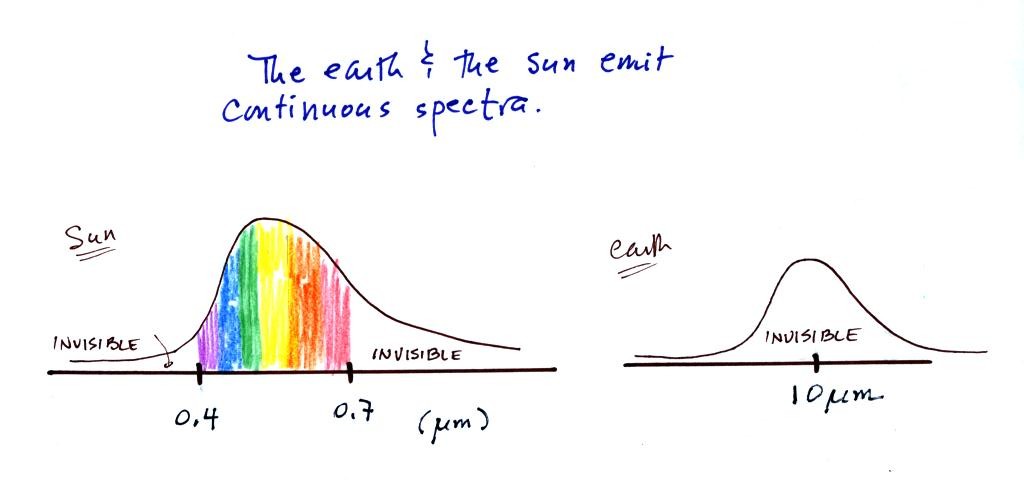
In some respects the earth and the sun behave just like the
tungsten filament in the light bulb used in the class
demonstration. Both the earth and the sun emit continuous
spectra. Part of the light emitted by the sun falls in the
visible part of the spectrum. If you were to look at the sun
(something you shouldn't do of course) with one of the diffraction
gratings handed out in class you would see all the colors of visible
light. There wouldn't be any gaps or colors missing. This
is shown again at the top of the figure below.
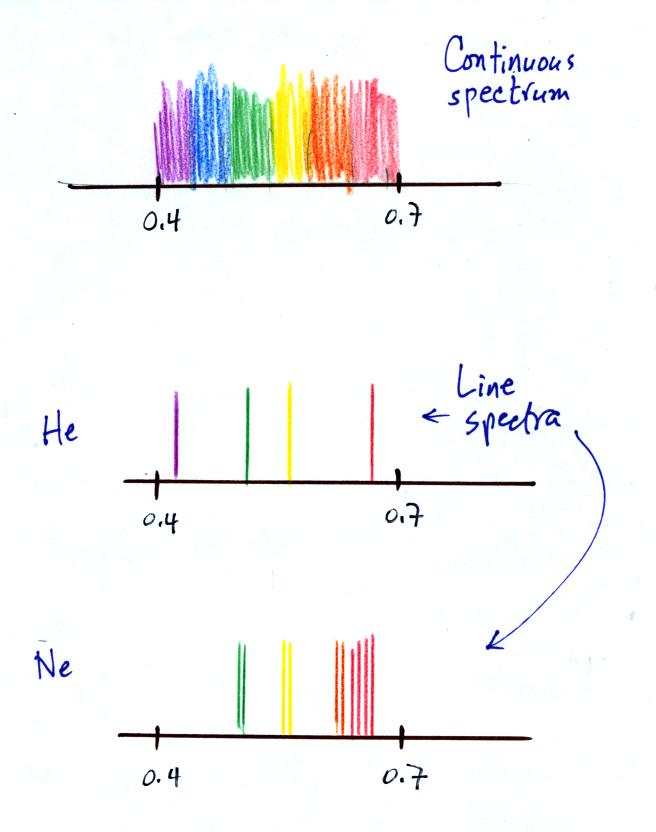
Gases behave differently. We looked at the visible light
emitted by helium (a high-voltage power supply was need to heat the
helium so that it would be hot enough to emit visible light).
Rather than a continuous spectrum, the helium emitted only certain
wavelengths. This is called a line spectrum. Helium
contains only two electrons and is a relatively simple atom and its
line spectrum was also fairly simple.
Neon (Ne) also emitted a line spectrum, thought there were more lines
at different wavelengths.
Finally we looked at the visible light emitted by molecular
nitrogen. In this case the spectrum was somewhere in between a
continous spectrum and a line spectrum. You were able to see
bands of colored light rather than clear line emitted by the nitrogen
(the bands are probably very closely spaced lines).
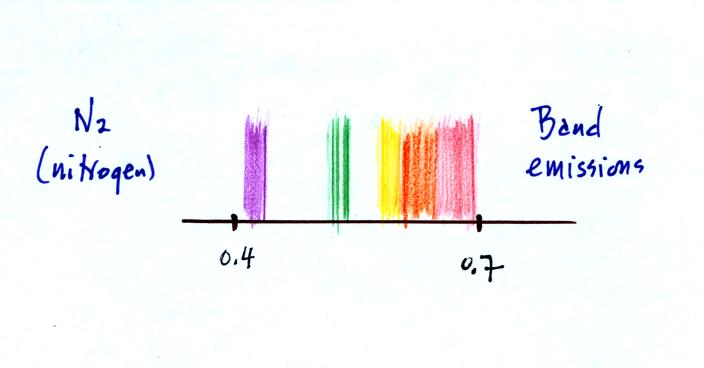
This is closer to being a continous spectrum but there were still
gaps, wavelengths without any emissions.
In the
same kind of way, air absorbs some wavelengths and transmits
others. We will need to worry about the filtering effect of the
atmosphere on ultraviolet, visible, and infrared light because all
three types of light are found in sunlight.


 This is really just a partial
list of some of the different
types of EM
radiation. In the top list, shortwave length and high energy
forms of EM radiation are on the left (gamma rays and X-rays for
example). Microwaves and radiowaves are longer wavelength, lower
energy forms of EM radiation.
This is really just a partial
list of some of the different
types of EM
radiation. In the top list, shortwave length and high energy
forms of EM radiation are on the left (gamma rays and X-rays for
example). Microwaves and radiowaves are longer wavelength, lower
energy forms of EM radiation.








